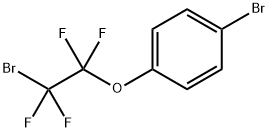
4-Bromophenoxytetrafluoroethyl bromide is a fluoroalkylbromide that is a radical source of the 4-bromophenoxytetrafluoroethyl moiety. Alternatively, it can also be selectively metallated at the fluorinated carbon with Turbo Grignard reagent at low temperatures resulting in a thermally unstable anion that can act as a nucleophilic fluoroalkylation reagent towards a wide variety of electrophiles, such as aldehydes, ketones or sulfonylimines.The aromatic bromide can be further subjected to Pd-catalyzed cross-coupling reactions to build molecular complexity.