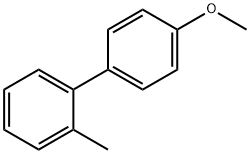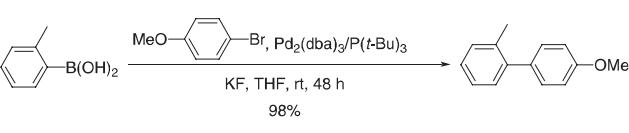
Open to the atmosphere, 4-bromoanisole (1.87 g, 10 mmol), o-tolylboronic acid (1.50 g, 11 mmol), KF (spray dried, dried in an oven overnight prior to use, 1.92 g, 33 mmol), and THF (10 mL) were added to a 100-ml round-bottomed Schlenk flflask equipped with a stir bar. The reaction system was flflushed with argon for about 5 min. P(t-Bu)3 (1.9 × 10–4 M stock solution in THF; 2.31 mL, 5.0 × 10–5 mmol) and Pd2(dba)3 (2.16 × 10–5 M stock solution in THF; 2.31 mL, 5.0 × 10–5 mmol) in THF were added sequentially. After 48 h at room temperature, the reaction mixture was diluted with ether or EtOAc, fifiltered through a pad of silica gel with copious washings, and then concentrated. The crude product was then purifified via column chromatography eluting with 5% ether in hexane to yield 1.94 g (98%) of 4-methoxy-2´-methylbiphenyl as a colorless liquid.