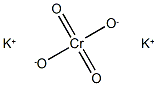
Potassium chromate
7789-00-6 Basic informationMore
- Product Name:Potassium chromate
- Synonyms: dipotassiumchromate(k2cro4) dipotassiummonochromate neutralpotassiumchromate potassiumchromate[k2(cro4)] potassiumchromate6 CHROMIUM(VI) ATOMIC SPECTR. STD. CONC. 1 .00 G CR(VI), AMP. CHROMIUM(VI) ATOMIC SPECTROSCOPY STANDA& POTASSIUM CHROMATE ACS REAGENT
- CAS:7789-00-6
- MF:CrK2O4
- MW:194.1903
- EINECS:232-140-5
- Mol File:7789-00-6.mol
-
USE
Potassium chromate can be used as analytical reagent, oxidant, mordant, metal rust inhibitor, and used for barium and silver trace analysis. it can also be used as raw materials of tanning and medicine industry and other chromium compounds. chromite fe(cro2)2 powder can be used as raw material and calcined with potassium hydroxide, limestone (dolomite) in the air, and then extracted with potassium sulfate solution to obtain potassium chromate.
Lastest Price from Potassium chromate manufacturers
- Potassium chromate
-

- US $0.00 /KG
- CAS:7789-00-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
- 2025-03-12
- Potassium chromate
-

- US $10.00 /KG
- CAS:7789-00-6
- Min. Order: 100KG
- Purity: 99%
- Supply Ability: 100 mt
- 2025-02-19
Browse by Nationality 7789-00-6 Suppliers >Global suppliers
Beijing (8) Shanghai (49) Tianjin (10) Chongqing (3) Sichuan (5) Guangdong (11) Zhejiang (8) Fujian (3) Hubei (33) Shandong (13) Henan (4) Hebei (7) Liaoning (9) Anhui (4) Jiangsu (13) Shaanxi (3) Xinjiang (1) Hong Kong (1) Other (1) Member (180) All (186)
Please select the suppliers
Recommend You Select Member Companies
Recommend You Select Member Companies
- Company Name:Meryer (Shanghai) Chemical Technology Co., Ltd.
- Tel:021-61259108 18621169109
- Products Intro:Product Name:PotassiuM chroMate
CAS:7789-00-6
Purity:99.5% Package:1kg;2.5kg Remarks:D70009 More... - CB Index:62
- Related Information:Catalog(40228)
- Company Name:Energy Chemical
- Tel:021-021-58432009 400-005-6266
- Products Intro:Product Name:PotassiuM chroMate
CAS:7789-00-6
Package:100g,500g More... - CB Index:61
- Related Information:Catalog(44733)
- Company Name:Adamas Reagent, Ltd.
- Tel:400-6009262 16621234537
- Products Intro:Product Name:Potassium Chromate
CAS:7789-00-6
Purity:>=99.5%AR Package:500g Remarks:G83043C More... - CB Index:59
- Related Information:Catalog(14103)
- Company Name:Shanghai Hanhong Scientific Co.,Ltd.
- Tel:021-54306202 13764082696
- Products Intro:Product Name:Potassium chromate
CAS:7789-00-6
Purity:AR,99.5% Remarks:RE01010122 More... - CB Index:64
- Related Information:Catalog(42958)
- Company Name:Shandong Xiya Chemical Co., Ltd
- Tel:4009903999 13355009207
- Products Intro:CAS:7789-00-6
Purity:AR Package:25g;100g;500g;1kg More... - CB Index:57
- Related Information:Catalog(18736)