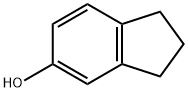
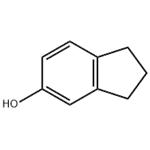
5-Indanol, mp 54–55 ℃, bp
255 ℃, is produced from indan [496-11-7] by
a sulfonation–alkali-fusion process. A
synthesis for 1,1,3,3-tetraalkylindanols was developed by Bayer, which involves
the reaction of isoolefins with secondary alkenylphenols (or compounds generating them
under the reaction conditions) at 100–250 ℃
and in the presence of acidic catalysts. An
example is the reaction of isobutene with bisphenol A in the presence of acid-activated
clays which yields 1,1,3,3-tetramethyl-5-indanol [53718-26-6], mp 115 ℃, bp (2.66 kPa)
156 ℃, in a yield of 80% of the theoretical,
and 4-tert-butylphenol as a coproduct.