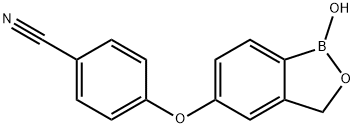
Crisaborole is a phosphodiesterase- 4 (PDE4) inhibitor that was approved by FDA on December 14, 2016 as Eucrisa, a topical treatment of for mild to moderate atopic dermatitis (AD) in patients aged two years and older. The drug, which was developed by Anacor Pharmaceuticals and later acquired and marketed by Pfizer, is delivered as a 2% ointment, which is applied topically. Its structure contains a boron atom, which facilitates skin penetration and binding to the bimetal center of the phosphodiesterase 4 enzyme. The unique employment of boron within the chemical structure of the drug is designed to enable selective engagement of PDE4 (an enzyme involved in the conversion of cAMP into AMP, which signals for downstream inflammatory events), effective penetration of the drug through human skin, and rapid clearance to limit systemic circulation.