Etanercept
한글명:Etanercept
카스 번호185243-69-0
상품명:Etanercept
CBNumberCB81373136
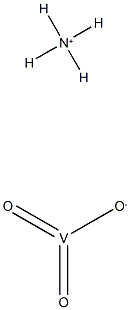
분자식H4NO3V
포뮬러 무게116.97816
MOL 파일185243-69-0.mol
| 유해 물질 데이터 | 185243-69-0(Hazardous Substances Data) |
그림문자(GHS)
-
그림문자(GHS)

-
신호 어
Warning
-
유해·위험 문구
H351:암을 일으킬 것으로 의심됨 (노출되어도 암을 일으키지 않는다는 결정적인 증거가 있는 노출경로가 있다면 노출경로 기재)
-
예방조치문구
P201:사용 전 취급 설명서를 확보하시오.
P202:모든 안전 조치 문구를 읽고 이해하기 전에는 취급하지 마시오.
P281:요구되는 개인 보호구를 착용하시오
P308+P313:노출 또는 접촉이 우려되면 의학적인 조치· 조언를 구하시오.
P405:밀봉하여 저장하시오.
P501:...에 내용물 / 용기를 폐기 하시오.
Etanercept 화학적 특성, 용도, 생산
-
개요
Etanercept is produced by recombinant DNA technology in a Chinese hamster ovary mammalian cell line and is the first biotechnology-derived drug to be introduced for the reduction of the signs and symptoms of moderately to severely active rheumatoid arthritis in patients who have not adequately responded to one or more of the synthetic DMARDs. It is a dimeric soluble form of the p75 TNFR capable of binding to two TNF molecules in the circulation. It consists of the extracellular ligand binding portion of the 75-kDa human TNFR fused to the Fc portion of human IgG1.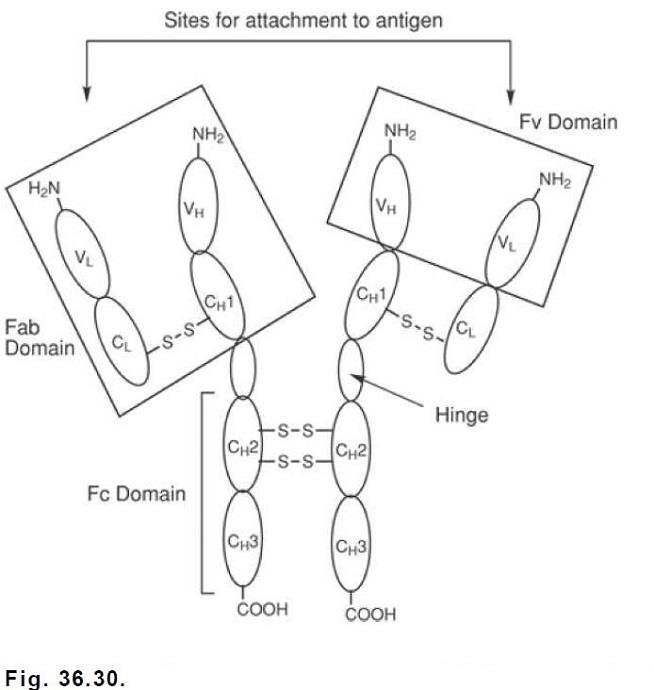
The Fc component of etanercept contains the CH2 domain, the CH3 domain, and the hinge region, but not the CH1 domain of IgG1. It consists of 934 amino acids and has an apparent molecular weight of approximately 150 kDa. Two TNFRs have been identified, a 75-kDa protein and a 55-kDa protein, that occur as monomeric molecules on cell surfaces and soluble forms in the blood. The biological activity of TNF requires its binding to either of the two cell surface TNFRs. Etanercept can bind specifically to two molecules of TNFα in the circulation, preventing its interaction with cell surface TNFRs. -
용도
To decrease signs and symptoms of rheumatoid arthritis.
Etanercept (Enbrel) is a soluble TNF receptor fusion protein. It is FDA approved for moderate to severe psoriasis at a dose of 50 mg twice weekly by subcutaneous injection for the initial 12 weeks, followed by a stepdown to 50 mg weekly for maintenance. The most common side effect is injection site reactions. Rare cases of serious infection, demyelinating disease, and congestive heart failure have been reported by postmarketing surveillance. -
Indications
Etanercept (Enbrel) is a recombinant fusion protein designed to block the action of TNF-α. The drug is composed of the extracellular ligand-binding portion of the 75-kilodalton human TNF receptor linked to the Fc portion of human IgG1. TNF-α is a cytokine thought to play a major role in the pathogenesis of a number of inflammatory skin diseases, including psoriasis. Etanercept binds soluble TNF-α, preventing it from binding to and activating receptors for TNF that are present on cell membranes. -
Pharmacology
Etanercept (Enbrel) is a soluble TNF receptor fusion protein. It is FDA approved for moderate to severe psoriasis at a dose of 50 mg twice weekly by subcutaneous injection for the initial 12 weeks, followed by a stepdown to 50 mg weekly for maintenance. -
Clinical Use
Etanercept is approved in the United States for the treatment of psoriatic arthritis and rheumatoid arthritis. Although etanercept has not been specifically approved for the treatment of the cutaneous manifestations of psoriasis, it significantly improves the skin lesions of patients with moderate to severe cutaneous psoriasis who have used it for psoriatic joint disease. -
부작용
The most common adverse reaction to etanercept is mild to moderate erythema, pain, or pruritus at the injection site (37%). Headaches and abdominal pain can also occur. New positive autoantibodies, such as antinuclear antibodies (ANA), anti-dsDNA antibodies, and anticardiolipin antibodies, can develop in patients treated with etanercept. Although there is so far no association between this and the development of autoimmune diseases or malignancies, long-term studies have yet to be done. Rare cases of pancytopenia may be associated with this drug. Although clinical trials showed no increased risk of infection with etanercept treatment, postmarketing reports of serious infections, sepsis, and associated fatalities exist. -
주의 사항
Etanercept therapy should not be initiated in patientswith active infection. If an infection develops in a persontaking etanercept, he or she should be closely monitored.If a serious infection or sepsis occurs, the drugshould be discontinued. Etanercept should be usedwith caution in individuals who have conditions predisposingthem to serious infection (e.g., uncontrolleddiabetes, hematological abnormalities). Data on druginteractions are limited. Live virus vaccines are contraindicatedbecause of the potential for secondarytransmission of the infection by the vaccine. Myelosuppressiveantirheumatic agents have been associatedwith pancytopenia in some patients treated with etanercept.
Etanercept 공급 업체
Global(66)Suppliers
-
전화+1-781-999-5354<br/>+1-00000000000
이메일 marketing@targetmol.com
-
Dideu Industries Group Limited
전화+86-29-89586680<br/>+86-15129568250
이메일 1026@dideu.com
-
전화+86-0571-85134551
이메일 sales@afinechem.com
-
Hefei Hirisun Pharmatech Co., Ltd
전화 +8615056975894
이메일 shawn@hirisunpharm.com
-
Hangzhou Huarong Pharm Co., Ltd.
전화571-86758373<br/>+8613588754946
이메일 sales@huarongpharm.com
-
전화 +8618058761490
이메일 info@gihichemicals.com
-
Wuhan Topule Biopharmaceutical Co., Ltd
전화 +8618327326525
이메일 masar@topule.com
-
Shanghai Acmec Biochemical Technology Co., Ltd.
전화+86-18621343501;<br/>+undefined18621343501
이메일 product@acmec-e.com
-
SHANGHAI KEAN TECHNOLOGY CO., LTD.
전화 +8613817748580
이메일 cooperation@kean-chem.com
-
Amadis Chemical Company Limited
전화571-89925085
이메일 sales@amadischem.com
1of2