Synthesis
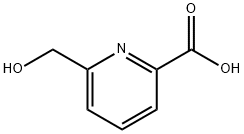
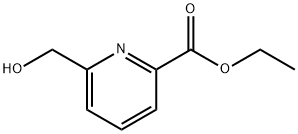
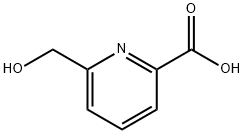
General procedure for the synthesis of 6-carboxypyridine-2-methanol from ethyl 6-hydroxymethylpyridine-2-carboxylate: ethyl 6-hydroxymethylpyridine-2-carboxylate (200 mg, 1.1 mmol) was dissolved in methanol (1 mL). Subsequently, 2 N NaOH aqueous solution (1 mL) was added slowly and the reaction mixture was stirred at room temperature for 1 hour. Upon completion of the reaction, the reaction mixture was acidified to pH=3 with 2 N HCl. The solvent was removed by concentration under reduced pressure, and the residue was dissolved in methanol and filtered. The filtrate was concentrated under reduced pressure to give 6-carboxypyridine-2-methanol (150 mg, 89% yield, white solid).1H NMR (400 MHz, CD3OD) δ: 5.05 (s, 2H), 8.34 (d, J=8.0 Hz, 1H), 8.47 (d, J=8.0 Hz, 1H), 8.73 (d, J=8.0 Hz, 1H).
References
[1] Zeitschrift fuer Naturforschung, B: Chemical Sciences, 1994, vol. 49, # 8, p. 1127 - 1136
[2] Patent: WO2004/113281, 2004, A1. Location in patent: Page 57
[3] Journal of the American Chemical Society, 1982, vol. 104, # 8, p. 2251 - 2257