ChemicalBook > CAS DataBase List > Fenticonazole nitrate
Fenticonazole nitrate
Fenticonazole nitrate
- CAS No.73151-29-8
- Chemical Name:Fenticonazole nitrate
- CBNumber:CB8481997
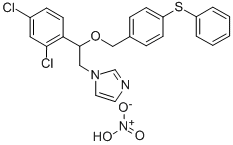
- Molecular Formula:C24H21Cl2N3O4S
- Formula Weight:518.41
- MOL File:73151-29-8.mol
Fenticonazole nitrate Property
- Melting point 135-137°C
- storage temp. Inert atmosphere,Room Temperature
- solubility Practically insoluble in water, freely soluble in dimethylformamide and in methanol, sparingly soluble in anhydrous ethanol
- form Solid
- pka 6.54(at 25℃)
- color White to Pale Yellow
- Merck 14,4005
- InChIKey FJNRUWDGCVDXLU-UHFFFAOYSA-N
- CAS DataBase Reference 73151-29-8(CAS DataBase Reference)
- FDA UNII 8V4JGC8YRF
- UNSPSC Code 41116107
- NACRES NA.77
Safety
- RTECS :NI4780000
- HS Code :2933.29.4300
- Toxicity :LD50 in mice, male rats, female rats (mg/kg): 1191, 440, 309 i.p.; all >3000 orally (Graziani)
-
Symbol(GHS)

- Signal wordWarning
- Hazard statements H315-H319
- Precautionary statements P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313
Fenticonazole nitrate Price
More Price(1)
- Brand: TCI Chemicals (India)
- Product number: F1008
- Product name : Fenticonazole Nitrate
- Purity:
- Packaging: 200MG
- Price: ₹8800
- Updated: 2022/05/26
- Buy: Buy
Fenticonazole nitrate Chemical Properties,Usage,Production
- Description Fenticonazole nitrate is an antifungal imidazole derivative reported to be more effective than other imidazole antifungals such as miconazole, clotrimazole and econazole. In addition to its potent, broad-spectrum fungicidal activity, fenticonazole is also active against some Gram-positive bacteria.
- Description Fenticonazole is an imidazole with antimicrobial activity. It is active against clinical isolates of Candida species, including C. albicans, C. parapsilosis, C. tropicalis, C. krusei, and C. guilliermondii with MIC values ranging from 0.12 to 32 μg/ml. It is also active against clinical isolates of bacteria, including B. fragilis, associated with bacterial vaginosis (MICs = ≤0.03-16 μg/ml), as well as bacteria, including S. aureus and S. epidermidis, associated with skin infections with MIC values ranging from 0.03 μg/ml or less to greater than 16 μg/ml. Fenticonazole inhibits fungal growth in a guinea pig model of experimental M. canis dermatomycosis (ED50 = 0.5%, applied topically).
- Chemical Properties Off-White Solid
- Originator Recordati (Italy)
- Uses Broad spectrum antimycotic; also active as antibacterial. Antifungal (topical).
- Definition ChEBI: A racemate comprising equimolar amounts of (R)- and (S)-fenticonazole nitrate. Used for the treatment of vaginal candidiasis.
-
Manufacturing Process
1-(2',4'-Dichlorophenyl)-2-chloroethanol:
49.5 g of sodium borohydride were added slowly and in small parts to a suspension of 233 g of 1-(1'-hydroxy-2'-chloroethyl)-2,4-dichlorobenzene in 1 liter of methanol stirred at room temperature. The solution thus obtained was stirred at room temperature for a further two hours, and it was then poured into 1 liter of 5 N hydrochloric acid cooled with ice. After extraction with ethyl acetate or chloroform, the extract was washed with water, with 1 N sodium hydroxide, then again with water until neutrality, and finally with a saturated sodium chloride solution. The extract was dried, the solvent evaporated off and 220 g of an oil were obtained. The oil solidified on standing and the solid 1-(2',4'-dichlorophenyl)-2-chloro-ethanol melted at 48-51°C.
1-(2',4'-Dichlorophenyl)-2-(N-imidazolyl)ethanol:
30 g of sodium were added to a solution of 88.5 g of imidazole in 600 ml of methanol; the solvent was then evaporated off. The residue was dissolved in 300 ml of dimethylformamide and heated to 115-120°C. To the solution so obtained was added, dropwise and under stirring, a solution of 225 g of 1- (2',4'-dichlorophenyl)-2-chloroethanol in 400 ml of dimethylformamide. The mixture was heated to 115-120°C and maintained at that temperature for 20 min and, after subsequent cooling to 40°C, 2500 ml of iced water were added under vigorous stirring. The product precipitated under stirring over a period of about two hours, the upper liquid was then decanted off, a further 2500 ml of water were added and, after standing, the whole was filtered. The precipitate thus obtained was dried and crystallized from toluene. 170 g of the 1-(2',4'-dichlorophenyl)-2-(N-imidazolyl)ethanol, melting at 134-135°C, was obtained.
METHOD 1:
A solution of 2.57 g of 1-(2',4'-dichlorophenyl)-2-(N-imidazolyl)ethanol in 10 ml of hexamethylphosphoramide was dropped at 25°C into a suspension of 0.52 g of sodium hydride (50% in oil) in 5 ml of hexamethylphosphoramide. When hydrogen emission was over, the salification was completed by heating for 1 hour at 50°C. After cooling to 25°C, 2.58 g of 1-chloromethyl-4- phenylthiobenzene were added. The temperature was raised to 50°C and maintained at that temperature for 12 hours. At the end of the reaction, the mixture was poured into 200 ml of water, the product was extracted with diethyl ether, the solvent was evaporated off and the residue was purified twice on a silica gel column, using ethyl acetate as eluant and testing the various fractions by TLC. The solvent was evaporated off the middle fractions to give 2.4 g of the 1-[2,4-dichloro-beta-[[p-(phenylthio)benzyl]oxy] phenethyl]imidazole as a yellowish oil, showing a single spot on TLC.
METHOD 2:
0.66 g of sodium hydride (50% in oil) were added at 20-30°C and under nitrogen atmosphere to 3.86 g of 1-(2',4'-dichlorophenyl)-2-(N-imidazolyl) ethanol in 15 ml of dimethylsulphoxide (dried on calcium hydride). The mixture was heated under stirring at 50-60°C until gas emission was over. After cooling to 20-25°C, 0.5 g of potassium iodide were added and slowly a solution of 3.51 g of 1-chloromethyl-4-phenylthiobenzene in 4 ml of dimethylsulphoxide was dropped in. The mixture was stirred at 20-25°C until addition of the 1-chloromethyl-4-phenylthiobenzene was over. The mixture was then poured into 150 ml of water and extracted with diethyl ether. To the etheric solution, after drying on anhydrous sodium sulphate, was added excess 4 N nitric acid solution in diethyl ether: the desired product precipitated as nitrate, an oil which solidified on standing. After standing for 20 hours, the etheric liquid was decanted off and the residue was crystallized from ethanol. The nitrate thus obtained, not completely pure, was dissolved in water and excess sodium carbonate was added in order to liberate the base which was then extracted with ethyl acetate. The base, obtained by filtration, was purified on a silica gel column using ethyl acetate as eluant. The combined fractions containing the desired product were evaporated to dryness. The residue was dissolved in diethyl ether, again transformed into the nitrate and crystallized from ethanol. Yield: 3.1 g of a white crystalline powder, melting at 136°C; λmax 252 nm (methanol). - brand name Lomexin
- Therapeutic Function Antifungal
Fenticonazole nitrate Preparation Products And Raw materials
Raw materials
Preparation Products
Global(292)Suppliers
-
Supplier:
Frapp's ChemicalNFTZ Co., Ltd.
- Tel:+86 (576) 8169-6106
- Email:sales@frappschem.com
- Country:China
- ProdList:880
- Advantage:50
-
Supplier:
Capot Chemical Co.,Ltd.
- Tel:+86-(0)57185586718<br/>+86-13336195806
- Email:sales@capot.com
- Country:China
- ProdList:29730
- Advantage:60
-
Supplier:
Beijing Cooperate Pharmaceutical Co.,Ltd
- Tel:010-60279497
- Email:sales01@cooperate-pharm.com
- Country:CHINA
- ProdList:1803
- Advantage:55
-
Supplier:
Henan Tianfu Chemical Co.,Ltd.
- Tel:+86-0371-55170693<br/>+86-19937530512
- Email:info@tianfuchem.com
- Country:China
- ProdList:21628
- Advantage:55
-
Supplier:
ATK CHEMICAL COMPANY LIMITED
- Tel:+undefined-21-51877795
- Email:ivan@atkchemical.com
- Country:China
- ProdList:33024
- Advantage:60
-
Supplier:
Shanxi Naipu Import and Export Co.,Ltd
- Tel:+86-13734021967<br/>+8613734021967
- Email:kaia@neputrading.com
- Country:China
- ProdList:1001
- Advantage:58
-
Supplier:
career henan chemical co
- Tel:+86-0371-86658258<br/>+8613203830695
- Email:sales@coreychem.com
- Country:China
- ProdList:29858
- Advantage:58
-
Supplier:
Xiamen AmoyChem Co., Ltd
- Tel:+86-86-5926051114<br/>+8615060885618
- Email:sales@amoychem.com
- Country:China
- ProdList:6383
- Advantage:58
-
Supplier:
TopScience Biochemical
- Tel:00852-68527855
- Email:info@itopbiochem.com
- Country:China Hong Kong
- ProdList:902
- Advantage:58
-
Supplier:
CONIER CHEM AND PHARMA LIMITED
- Tel: +8618523575427
- Email:sales@conier.com
- Country:China
- ProdList:49732
- Advantage:58
73151-29-8, Fenticonazole nitrateRelated Search:
- Sulfur & Selenium Compounds
- Pharmaceuticals
- Intermediates & Fine Chemicals
- Heterocycles
- Aromatics
- Inhibitors
- 抑制剂
- 配体家族
- 药靶配体
- 化学试剂
- 医药、农药及染料中间体
- 原料
- 化工中间体工业原料
- 药物杂质及中间体
- 医药原料药
- 科研试剂
- 原料药
- 医药化工类
- 医药 抗真菌药
- 抗真菌药
- 医药中间体
- 小分子抑制剂,天然产物
- 医药原料
- 药物
- 抗真菌感染药物
- 抗病源性微生物药
- 非抗生素类抗感染原料药
- C24H21Cl2N3O4S
- C24H20Cl2N2OSHNO3
- 724729-26-6
- 硝酸芬替康唑,10 MM DMSO 溶液
- 硝酸芬替康唑标准品
- 1-(2-(2,4-二氯苯基)-2-((4-(苯硫基)苄基)氧基)乙基)-1H-咪唑硝酸盐
- 硝酸芬替康唑/硝酸抑霉唑
- 硝酸芬替康唑 EP标准品
- 硝酸芬康唑费用
- 酸芬替康唑
- 硝酸抑霉唑
- 硝酸芬替康唑
- 硝酸芬康唑
- 1-[2-(2,4-二氯苯基)-2-[(4-苯基硫苯基)甲氧基]乙基]咪唑硝酸盐
- 1-[2-(2,4-二氯苯基)-2-[[4-(苯硫代)苯基]甲氧基]乙基]-1H-咪唑硝酸盐
- 1-[2-(2,4-二氯)苯基-2-(4-苯硫基)苯甲氧基]乙基-1H-咪唑硝酸盐
- 氟达拉滨磷酸
- 73151-29-8
- Fenticonazole Nitrate, 10 mM in DMSO
- Fenticonazole Nitrate 1-(2-(2,4-dichlorophenyl)-2-((4-(phenylthio)benzyl)oxy)ethyl)- 1H-imidazole nitrate
- Fenticonazole Nitrate EP
- 1-[2-(2,4-dichlorophenyl)-2-[[4-(phenylthio)phenyl]methoxy]ethyl]-1H-imidazole,nitrate (1:1)
- Fenticonazole nitrate CRS
- ((2R,3R,4S,5R)-5-(6-amino-2-fluoro-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl)methyl dihydrogen phosphate
- TIANFU-CHEM - Fenticonazole nitrate
- 1-(2-(2,4-Dichlorophenyl)-2-((4-(phenylthio)benzyl)oxy)ethyl)-1H-imidazole nitrate
- Fenticonazole nitrate USP/EP/BP
- FenticonazoleNitrate>
- 1-[2-(2,4-dichlorophenyl)-2-[[4-(phenylthio)phenyl]methoxy]ethyl]-1H-imidazolium nitrate
- FENTICONAZOLE NITRATE
- 1-(2-(2,4-DICHLOROPHENYL)-2-((4-(PHENYLTHIO)BENZYL)OXY)ETHYL)-1H-IMIDAZOLE