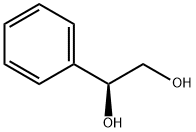
Chemical Properties
(S)-(+)-1-Phenyl-1,2-ethanediol is white to light yellow crystal powde
Uses
(S)-(+)-1-Phenyl-1,2-ethanediol used as a chiral building block in organic synthesis, used in the preparation of chiral 2-amino-1-phenylethanols as pharmaceutical intermediates.
Uses
Extremely valuable and versatile alcohol used as a chiral building block in organic synthesis, used in the preparation of chiral 2-amino-1-phenylethanols as pharmaceutical intermediates.
Biosynthesis
Terminal vicinal diols are important chiral building blocks and intermediates in organic synthesis. Reduction of α-hydroxy ketones provides a straightforward approach to access these important compounds. It has been found that asymmetric reduction of a series of α-hydroxy aromatic ketones and 1-hydroxy-2-pentanone, catalyzed by Candida magnolia carbonyl reductase (CMCR) with glucose dehydrogenase (GDH) from Bacillus subtilis for cofactor regeneration, afforded 1-aryl-1,2-ethanediols ((S)-(+)-1-Phenyl-1,2-ethanediol) and pentane-1,2-diol, respectively, in up to 99 % ee.[1]
Purification Methods
Purify the diol by recrystallisation from *C6H6/ligroin and sublime it at 1-2mm. [Arpesella et al. Gazetta 85 1354 1955, Prelog et al. Helv Chim Acta 37 221 1954, Beilstein 6 IV 5939.]
References
[1] Highly Efficient Synthesis of Optically Pure (S)-1-phenyl-1,2-ethanediol by a Self-Sufficient Whole Cell Biocatalyst. DOI:
10.1002/open.201500045