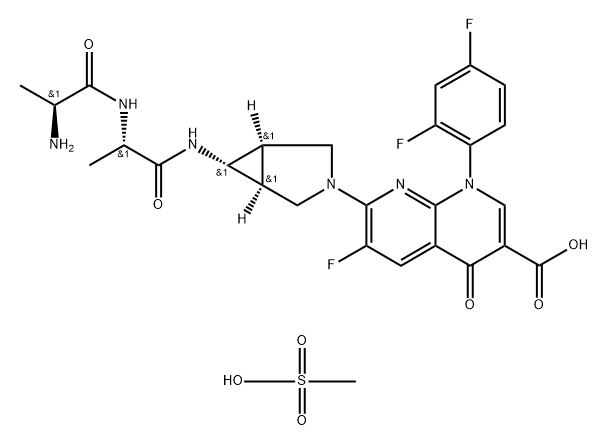
Uses
Alatrofloxacin Mesylate is the parental prodrug of Trovafloxacin (T893000), on phagocytic, anti-inflammatory and immunomodulation events of human THP-1 monocytes.
Manufacturing Process
The dipolar cycloaddition of ethyldiazoacetate to the protected dihydropyrrole
(with carbobenzyloxy (CBZ) protecting group) gives the fused pyrrazolidine-
(CBZ-3-ethylperoxy-1,3a,4,5,6,6a-hexahydro-pyrrolo{3,4-c}pyrazole).
Pyrolysis results in loss of nitrogen and formation of the cyclopyrrolidine ring.
The ester is then saponified to the corresponding carboxylic acid (CBZ-6-azabicyclo[
3.1.0]hexene-3-carboxylic acid).This acid undergoes a version of the Curtius rearrangement when treated with diphenylphosphinous azide to afford
transient isocyanate. That reactive function adds tert-butanol from the
reaction medium to afford the product as its butoxycarbonyl (BOC) derivative.
CBZ-protecting group is removed by catalytic hydrogenation on Pd to afford
the BOC-protected secondary amine - BOC-6-aza-bicyclo[3.1.0]hex-3-ylamine.
In a standard quinolone reaction, this amine is then used to displace the more
reactive fluorine at 7-position in the quinolone - 1-(2,4 difluoro-phenyl)-6,7-
difluoro-4-oxo-1,4-dihydro[1,8]naphthylridine-3-carboxylic acid ethyl ester.
Treatment of the displacement product with hydrogen chloride cleaves the
BOC protecting group to afford the antibiotic trovafloxacin. The peptide-like
alanylalanylamide derivative, alatrofloxacin can in principle be prepared by
reaction of the ester precursor of trovafloxacin with alanylalanine followed by
saponification and treatment by methane sulphonic acid (mesylate).