Synthesis
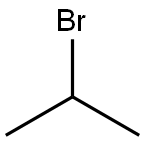
The general procedure for the synthesis of 3-iodo-1-isopropyl-1H-pyrazolo[3,4-d]pyrimidin-4-amine from 3-iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine (5 g, 19.2 mmol, 1.0 eq.) and 2-bromopropane (1.9 mL, 20.1 mmol, 1.05 eq.) was as follows: 3-Iodo-1-isopropyl-1H-pyrazolo[3,4-d]pyrimidin-4-amine was placed in a three-necked flask. d]pyrimidin-4-amine in a three-necked flask with 40 mL of DMF and potassium carbonate (5.3 g, 38.4 mmol, 2.0 eq.). The mixture was replaced with nitrogen 3 times and then 2-bromopropane was added. The reaction mixture was heated to 80 °C and the reaction progression was monitored by TLC after 4 hours of reaction. Upon completion of the reaction, DMF was removed by distillation under reduced pressure. the residue was extracted three times with a solvent mixture of dichloromethane (DCM) and water, the DCM layers were combined and evaporated to dryness. The product was purified by recrystallization from a solvent mixture of ethyl acetate (EA) and petroleum ether (PE) (1:3, v/v) to afford the target compound in the form of a pale yellow powder. The product in the recrystallized mother liquor was further purified by column chromatography to give 5.4 g of product in 92.8% yield. The product was characterized by 1H NMR (400 MHz, DMSO-d6): δ 8.19 (s, 1H), 4.99-4.93 (m, 1H), 1.42 (d, J=6.7 Hz, 6H). Mass spectrum (ESI) showed m/z: 304.0 [M+H].
References
[1] Patent: US2017/305920, 2017, A1. Location in patent: Paragraph 0147
[2] Patent: US2015/225407, 2015, A1. Location in patent: Paragraph 0407
[3] Patent: US2007/293516, 2007, A1. Location in patent: Page/Page column 12; 16
[4] Patent: WO2010/6086, 2010, A2. Location in patent: Page/Page column 79
[5] Patent: US2016/789, 2016, A1. Location in patent: Paragraph 0322
![3-Iodo-1-isopropyl-1H-pyrazolo[3,4-d]pyrimidin-4-amine Structure](https://www.chemicalbook.com/CAS/GIF/862730-04-9.gif)
![3-Iodo-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/151266-23-8.gif)
![3-Iodo-1-isopropyl-1H-pyrazolo[3,4-d]pyrimidin-4-amine](/CAS/GIF/862730-04-9.gif)