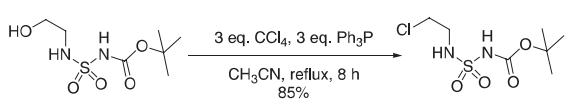
Synthesis
A solution of BOC-sulfamoylaminoalcohol (5.35 mmol), triphenylphosphine (16.05 mmol), and CCl4 (16.05 mmol) in anhydrous acetonitrile (100 mL) was reflfluxed for 8 h. After cooling to room temperature, the solution was concentrated in vacuo. The residue was triturated with diethyl ether (3 × 150 mL). Triphenylphosphine oxide, which precipitates in the combined organic layers, was removed by fifiltration. The fifiltrate was concentrated and the residue purifified on silica gel (CH2Cl2) to afford N1-BOC,N3-(2-chloroethyl) sulfamide with 85% yield.
Reference: Regaïnia, Z.; Abdaoui, M.; Aouf, N.-E.; Dewynter, G.; Montero, J.-L. Tetrahedron 2000, 56, 381–387.