Synthesis
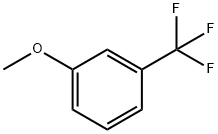
The general procedure for the synthesis of 2-(3-methoxy-5-(trifluoromethyl)phenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolanes from m-trifluoromethyl anisole and bis(pinacolato)boronic acid esters was as follows: to a flame-dried 100 mL Schlenk tube were added sequentially bis(pinacolato)diboron (350 mg, 1.38 mmol), [Ir( COD)Cl]2 (12 mg, 0.018 mmol), sodium methanol (5 mg, 0.09 mmol), and 4,4'-di-tert-butyl-2,2'-bipyridine (8 mg, 0.03 mmol). The flask was evacuated and filled with argon for protection, followed by the addition of 3-trifluoromethylanisole (2.5 mL). The flask was again evacuated (complete vacuum for 2 min), then sealed under vacuum and placed in a 90 °C oil bath for 96 h of reaction. Upon completion of the reaction, the reaction mixture was transferred to a round bottom flask using ethyl acetate and purified by Kugelrohr distillation. The resulting product was a viscous oil and was distilled at 120°C for 10 min. Final isolation gave 497 mg (1.65 mmol, 60% yield) of the target compound. The 1H NMR (400 MHz, CDCl3) data of the product were as follows: δ 7.62 (s, 1H), 7.43 (s, 1H), 7.18 (s, 1H), 3.82 (s, 3H), 1.32 (s, 12H).
References
[1] Journal of the American Chemical Society, 2018,
[2] Tetrahedron, 2008, vol. 64, # 29, p. 6824 - 6830
[3] Patent: US2016/168151, 2016, A1. Location in patent: Paragraph 0241
[4] Angewandte Chemie - International Edition, 2002, vol. 41, # 16, p. 3056 - 3058
[5] Organic Letters, 2007, vol. 9, # 5, p. 757 - 760
![2-(3-Methoxy-5-trifluoromethyl-phenyl)-4,4,5,5-tetramethyl-[1,3,2]dioxaborolane Structure](https://www.chemicalbook.com/CAS/GIF/479411-93-3.gif)



![2-(3-Methoxy-5-trifluoromethyl-phenyl)-4,4,5,5-tetramethyl-[1,3,2]dioxaborolane](/CAS/GIF/479411-93-3.gif)