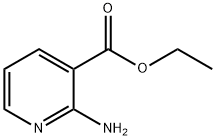
Synthesis
General procedure for the synthesis of ethyl 2-amino-5-bromonicotinate from ethyl 2-aminonicotinate: to a stirred solution of ethyl 2-aminonicotinate (15 g; 90.36 mmol; 1 eq.) in anhydrous THF (150 mL) was added N-bromosuccinimide (NBS) (16 g; 90.36 mmol; 1 eq.) in batches at 0 °C. The resulting mixture was stirred at 23 °C for 18 hours. Upon completion of the reaction, the mixture was poured into ice-cold saturated aqueous NaHCO3 solution and the organic components were extracted with ethyl acetate (3 x 200 mL). The combined organic layers were washed with brine solution, dried over anhydrous sodium sulfate, filtered and concentrated to dryness under reduced pressure to afford ethyl 2-amino-5-bromonicotinate (22 g, 100%) as an off-white solid. The product was confirmed by 1H NMR (DMSO-d6): δ 8.29 (d, 1H, J = 3Hz), 8.12 (d, 1H, J = 2Hz), 7.31 (s, 2H), 4.29 (q, 2H, J = 7Hz), 1.30 (t, 3H, J = 7Hz).The LCMS analysis resulted in: m/z = 245.0 [M + H]+, 247.0 [M + 2H]+, retention time (RT) = 3.34 min (program P1, column W).
References
[1] Patent: WO2015/95128, 2015, A1. Location in patent: Paragraph 0239; 0242; 0243
[2] European Journal of Medicinal Chemistry, 2018, vol. 158, p. 236 - 246
[3] Patent: CN104402881, 2016, B. Location in patent: Paragraph 0022; 0023
[4] Patent: WO2007/67416, 2007, A2. Location in patent: Page/Page column 64; 134