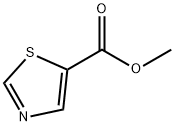
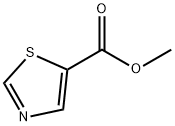
Synthesis
The general procedure for the synthesis of methyl 5-thiazolecarboxylate from methyl 2-aminothiazole-5-carboxylate was as follows: a mixture of 2.25 g of isoamyl nitrite with 10 mL of dioxane was stirred at 80 °C under nitrogen protection. Subsequently, a 20 mL dioxane solution containing 1.23 g of methyl 2-aminothiazole-5-carboxylate (g-3) was slowly added dropwise. After the dropwise addition was completed, the reaction mixture was refluxed for 2 hours. After the reaction was completed, it was cooled to room temperature and diluted by adding 30 mL of ethyl acetate. The organic phase was washed with saturated saline, dried over anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The crude product was purified by silica gel column chromatography to give 0.54 g (48% yield) of the target product methyl 5-thiazolecarboxylate (g-4).