Description
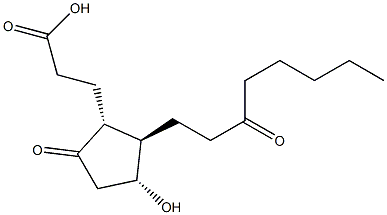
A common metabolic pathway for several prostaglandins (PG), including PGE
2, involves the reduction of the double bond between C-13 and C-14 and oxidation of the hydroxyl group at C-15, producing 13,14-dihydro-15-keto PGs. The removal of four carbons at the α-terminus and oxidation of the terminal ω-carbon produces the abundant urinary metabolites, including tetranor PGEM.
1 13,14-dihydro-15-keto-tetranor PGE
2 is a potential metabolite of PGE
2. It would be produced from the known metabolite 13,14-dihydro-15-keto PGE
2 , which is known to have a short plasma half-life.
2,3,4
References
1. Hamberg, M.
Inhibition of prostaglandin synthesis in man Biochem. Biophys. Res. Commun. 49(3),720-726(1972).
2. Bothwell, W., Verburg, M., Wynalda, M., et al.
A radioimmunoassay for the unstable pulmonary metabolites of prostaglandin E1 and E2: An indirect index of their in vivo disposition and pharmacokinetics J. Pharmacol. Exp. Ther. 220(2),229-235(1982).
3. Fitzpatrick, F.A., Aguirre, R., Pike, J.E., et al.
The stability of 13,14-dihydro-15 keto-PGE2 Prostaglandins 19(6),917-931(1980).
4. Granstr?m, E., Hamberg, M., Hansson, G., et al.
Chemical instability of 15-keto-13,14-dihydro-PGE2: The reason for low assay reliability Prostaglandins 19(6),933-945(1980).