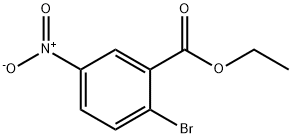
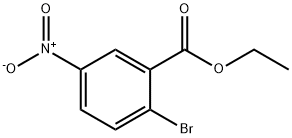
Ethyl 2-broMo-5-nitrobenzoate
- Product NameEthyl 2-broMo-5-nitrobenzoate
- CAS208176-31-2
- CBNumberCB62549789
- MFC9H8BrNO4
- MW274.07
- MDL NumberMFCD12025203
- MOL File208176-31-2.mol
- MSDS FileSDS
Chemical Properties
| Density | 1.594 |
| storage temp. | Sealed in dry,Room Temperature |
Ethyl 2-broMo-5-nitrobenzoate Price
| Product number | Packaging | Price | Product description | Buy |
|---|---|---|---|---|
| AK Scientific W4368 | 10g | $150 | Ethyl2-bromo-5-nitrobenzoate |
Buy |
| Matrix Scientific 075816 | 1g | $278 | Ethyl 2-bromo-5-nitrobenzoate 95+% |
Buy |
| AK Scientific W4368 | 100g | $589 | Ethyl2-bromo-5-nitrobenzoate |
Buy |
| Matrix Scientific 075816 | 5g | $700 | Ethyl 2-bromo-5-nitrobenzoate 95+% |
Buy |
| American Custom Chemicals Corporation HCH0314468 | 1G | $882.42 | ETHYL-2-BROMO-5-NITRO-BENZOATE 95.00% |
Buy |
Ethyl 2-broMo-5-nitrobenzoate Chemical Properties,Usage,Production
Synthesis

64-17-5
943-14-6
208176-31-2
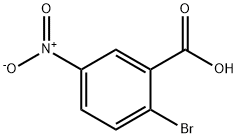
General procedure for the synthesis of ethyl 2-bromo-5-nitrobenzoate from ethanol and 2-bromo-5-nitrobenzoic acid: To a solution of 2-bromo-5-nitrobenzoic acid (1.50 g, 6.12 mmol) in methylene chloride (10 mL) was added oxaloyl chloride (1.10 g, 12.24 mmol), and the resultant solution was stirred for 30 min at room temperature. The reaction mixture was concentrated and the residue was dissolved in ethanol and the resulting solution was continued to be stirred for 2 hours at room temperature. Subsequently, the reaction solution was concentrated and the residue was dissolved in ethyl acetate. Water was added to this solution and the organic layer was washed with brine, separated, dried, filtered and concentrated to give 1.0 g of the target product ethyl 2-bromo-5-nitrobenzoate. The structure of the product was confirmed by 1H NMR (300 MHz, DMSO-d6): δ 8.50 (d, J = 3.0 Hz, 1H), 8.27-8.23 (dd, J = 3.0, 8.7 Hz, 1H), 8.06-8.03 (d, J = 8.7 Hz, 1H), 4.41-4.34 (q, J = 6.9 Hz, 2H), 1.37 -1.32 (t, J = 7.2 Hz, 3H).
References
[1] Patent: WO2014/167444, 2014, A1. Location in patent: Page/Page column 66Preparation Products And Raw materials
Ethyl 2-broMo-5-nitrobenzoate Supplier
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| +86-(0)57185586718; +8613336195806 |
sales@capot.com | China | 29640 | 60 | |
| 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 | |
| +8618523575427 | sales@conier.com | China | 49977 | 58 | |
| +86-021-021-50872116 +8613122107989 |
contact@fuxinpharm.com | China | 10000 | 58 | |
| +86-0086-577-64498589 +86-15355981851 |
sales@win-winchemical.com | China | 14304 | 58 | |
| +86-0519-89882563 +86-18961279162 |
andy@anxuanchem.com | China | 4327 | 58 | |
| +86-0551-65418684 +8618949823763 |
sales@tnjchem.com | China | 25356 | 58 | |
| +86-0571-85585865 +8613336024896 |
CB@aromsyn.com | China | 19183 | 58 | |
| +86-571-87711850 | market18@leapchem.com | China | 43333 | 58 | |
| tp@aladdinsci.com | United States | 52923 | 58 |
The What'sApp is temporarily not supported in mainland China