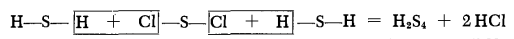Chemical Properties
Bright light-yellow liquid similar to olive
oil, pungent odor. Somewhat less easily decomposed than H2S2 and
H2S3; like these, it is converted on prolonged standing into
homologs richer in sulfur. No definite melting point; solidifies at
about —85°C to a white, glassy mass which gradually softens over
a fairly wide temperature range when heated. Cracked by heating
to lower hydrogen sulfides, particularly H3S. d (20°C) 1.582.
Synthesis
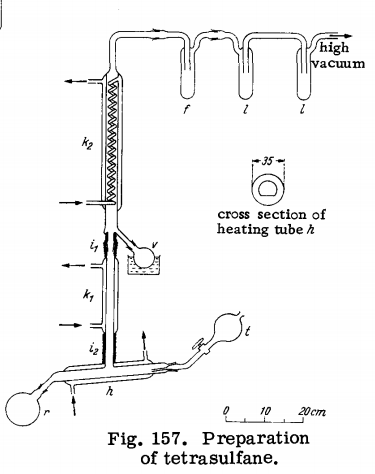
Tetrasulfane is obtained from crude sulfane in the apparatus
shown in Fig. 157. The modified Utzinger tube h, the lower part of which is
flattened to increase the vaporization surface as much as possible
(see cross section), is surrounded by a heating jacket and carries
a separatory funnel t and a 500-ml. flask r. The attached condenser K1 and the spiral condenser K 2 (which is identical to that
used in trisulfane purification and has a toroidal expansion and an
outlet to the receiver u) are provided with separate constant-temperature circulating systems. Again, condenser hs
is connected
to trap / , which is immersed in a Dry Ice-methanol mixture.
Trap / adjoins double trap I, cooled with liquid nitrogen, which is
connected to a high-vacuum pump. The connecting sections of the
condensers are covered with asbestos insulation^ and£a.