Synthesis
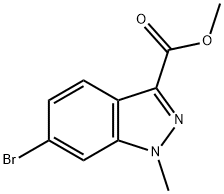
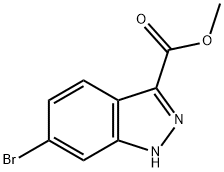
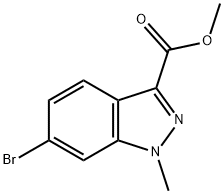
Intermediate Preparation 411B; Methyl 6-bromo-1-methyl-1H-indazole-3-carboxylate; To a suspension of methyl 6-bromo-1H-indazole-3-carboxylate (0.50 g, 1.96 mmol) and potassium carbonate (1.28 g, 9.29 mmol) in acetonitrile (15 mL) was added methiodine (1.31 g, 9.29 mmol). The reaction mixture was stirred at room temperature for 15 hours. After completion of the reaction, the solvent was removed under vacuum and the residue was diluted with water (15 mL) and extracted with ethyl acetate (3 x 10 mL). The organic phases were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by fast column chromatography using 8:2 hexane/ethyl acetate as eluent to afford 0.35 g (70% yield) of methyl 6-bromo-1-methylindazole-3-carboxylate as an off-white solid.1H NMR (400 MHz, CDCl3) δ 4.03 (s, 3H), 4.14 (s, 3H), 7.42 (dd, J = 8.4, 1.2 Hz, 1H), 7.66 (s, 1H), 8.08 (d, J = 8.8 Hz, 1H).
References
[1] Patent: WO2009/12125, 2009, A1. Location in patent: Page/Page column 38
[2] Patent: WO2011/20615, 2011, A1. Location in patent: Page/Page column 85; 86
[3] Patent: WO2018/177781, 2018, A1. Location in patent: Page/Page column 82-83