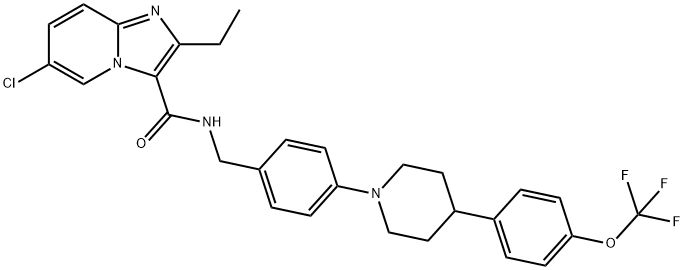
Telacebec (Q203) is efficacious in a mouse model of tuberculosis at a dose less than 1 mg per kg body weight. Telacebec displays pharmacokinetic and safety profiles compatible with once-daily dosing. Telacebec has a bioavailability of 90% and a terminal half-life of 23.4 h. The volume of distribution is moderate (5.27 l per kg body weight), and the systemic clearance is low (4.03 mL/min per kg). After 4 weeks of treatment, reductions of 90%, 99% and 99.9% in M. tuberculosis H37Rv bacterial load is observed in the groups treated with Telacebec at 0.4, 2 and 10 mg per kg body weight, respectively[1].