Synthesis
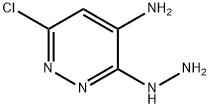
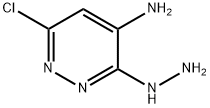
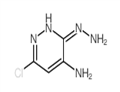
Step 112.2: Synthesis of 6-chloro-3-hydrazinopyridazin-4-amine
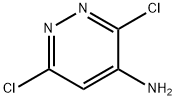
To a stirred suspension of 3,6-dichloropyridazin-4-amine (1.49 g, 9.09 mmol) in ethanol (15 mL) was slowly added hydrazine hydrate (11.04 mL, 227 mmol). The reaction mixture was heated to 100 °C and stirred for 3 hours. Upon completion of the reaction, the mixture was cooled to room temperature and subsequently concentrated under reduced pressure to remove the solvent. The crude product was ground with deionized water (25 mL) to afford the title compound 6-chloro-3-hydrazinopyridazin-4-amine (478 mg, 3 mmol, 33% yield) as a yellow solid.
Analytical data:
Retention time (t R): 0.24 min (LC-MS method 2);.
Positive ion mode electrospray ionization mass spectrometry (ESI-MS): m/z 160 [M + H]+;
Negative ion mode electrospray ionization mass spectrometry (ESI-MS): m/z 158 [M-H]-.
References
[1] Journal of Medicinal Chemistry, 2014, vol. 57, # 6, p. 2807 - 2812
[2] Patent: US2014/349990, 2014, A1. Location in patent: Paragraph 1053; 1054
[3] Patent: WO2014/191896, 2014, A1. Location in patent: Page/Page column 218
[4] Patent: US4728355, 1988, A