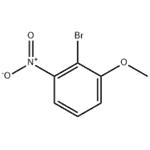
Synthesis
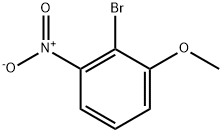
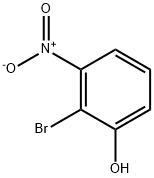
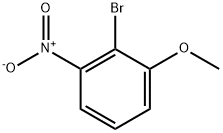
General procedure for the synthesis of 2-bromo-3-nitroanisole from 2-bromo-3-nitrophenol (3.1 g, 14.2 mmol) and iodomethane (2.6 mL, 42.5 mmol): 2-bromo-3-nitrophenol was dissolved in N,N-dimethylformamide (DMF, 20 mL), to which was added ground cesium carbonate (5.58 g, 17.1 mmol), followed by the addition of iodomethane. The reaction mixture was stirred at room temperature overnight. After completion of the reaction, DMF was removed by rotary evaporator and the residue was dissolved in ether (200 mL) and washed sequentially with water (200 mL) and saturated sodium chloride solution (4 x 100 mL). The organic phase was dried with anhydrous magnesium sulfate, filtered and concentrated to give the crude product 2-bromo-3-nitroanisole (94% yield, 3.1 g) as an orange solid. Mass spectrometry (MS) analysis showed m/z 234 ([M+2H]+). High performance liquid chromatography (HPLC, TFA system, 220 nm detection) showed a product purity of 98%.
References
[1] Patent: WO2004/103996, 2004, A1. Location in patent: Page 40-41
[2] Patent: WO2006/7700, 2006, A1. Location in patent: Page/Page column 62
[3] Patent: US2006/19905, 2006, A1. Location in patent: Page/Page column 24
[4] Patent: WO2006/85, 2006, A1. Location in patent: Page/Page column 75-76
[5] Patent: WO2007/9227, 2007, A1. Location in patent: Page/Page column 35