Synthesis
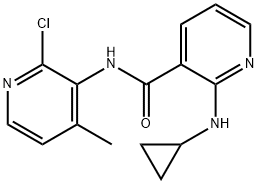
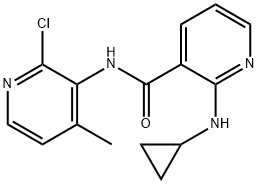
Example 7: Synthesis of N-(2-chloro-4-methyl-3-pyridinyl)-2-cyclopropylamino-3-pyridinecarboxamide (VII)
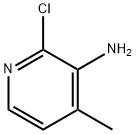
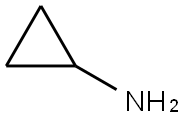
In an autoclave, 2-chloro-N-(2-chloro-4-methyl-3-pyridinyl)-3-pyridinecarboxamide (III; 1.0 kg, 3.5587 mol) was dissolved in toluene (6.0 liters). Cyclopropylamine (0.813 kg, 14.23 mol) was then added and the reaction mixture was heated at 125 °C to 130 °C and 15 psi pressure for 21 hours. After completion of the reaction, the mixture was cooled to 25 °C to 30 °C and washed with distilled water. After separating the aqueous layer, the aqueous layer was extracted with ethyl acetate (2.0 liters). The organic layers (toluene and ethyl acetate layers) were combined and concentrated under reduced pressure to give the target product. Yield: 0.857 kg, yield: 79.64%, HPLC purity: 98.14%.