Synthesis
Step 1: Synthesis of 6-bromo-2-methyloxazolo[5,4-b]pyridine
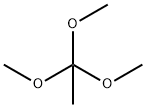
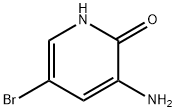
3-Amino-5-bromopyridin-2-ol (1.00 g, 5.29 mmol) was mixed with trimethyl orthoacetate (5 mL) and placed in a microwave reactor and the reaction was stirred at 120 °C for 2 hours. After completion of the reaction, the reaction mixture was concentrated and purified by silica gel column chromatography (eluent: petroleum ether/ethyl acetate = 5:1 to 1:1) to afford 550 mg (49% yield) of 6-bromo-2-methyloxazolo[5,4-b]pyridine as a white solid.
LC-MS: m/z 212.8 [M + H]+.
1H NMR (400 MHz, CDCl3) δ [ppm]: 8.36 (d, J = 2.1 Hz, 1H), 8.08 (d, J = 2.1 Hz, 1H), 2.69 (s, 3H).
![6-BROMO-2-METHYL-OXAZOLO[5,4-B]PYRIDINE Structure](https://www.chemicalbook.com/StructureFile/ChemBookStructure21/GIF/CB3816028.gif)
![6-BROMO-2-METHYL-OXAZOLO[5,4-B]PYRIDINE](/StructureFile/ChemBookStructure21/GIF/CB3816028.gif)