Synthesis
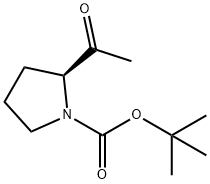
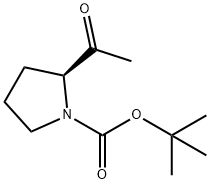
Step B: tert-butyl (S)-2-methoxymethylcarbamoylpyrrolidine-1-carboxylate (1.29 g, 4.99 mmol) obtained in Step A was dissolved in an appropriate amount of dry solvent, and the reaction mixture was cooled to -78 °C. Methylmagnesium bromide Grignard reagent (12 mL, 15 mmol) was slowly added under nitrogen protection, keeping the temperature at -78 °C. After addition, the reaction mixture was gradually warmed to room temperature and stirred for 1 hour. Upon completion of the reaction, the reaction mixture was again cooled to -78 °C and the reaction was terminated by slow addition of saturated aqueous ammonium chloride solution. The reaction mixture was extracted with ethyl acetate (EtOAc), the organic phases were combined and dried with anhydrous magnesium sulfate (MgSO4). The solvent was removed by concentration under reduced pressure to give the crude product. Purification by column chromatography (eluent: ethyl acetate/hexane = 1/1) afforded the target product (S)-tert-butyl 2-acetylpyrrolidine-1-carboxylate (1.03 g, 97% yield).