Manufacturing Process
(A) Preparation of dl-N-(γ-Chloromercuri-β-Methoxy)-Propylcamphoramic Acid: A suspension of 31.9 g (= 0.10 M) of mercuric acetate in 25 ml of methanol is stirred for 30 minutes at room temperature in a 4-necked flask equipped with stirrer, dropping funnel, drying tube and thermometer. To this suspension is added dropwise and with stirring, a solution of 23.9 g (= 0.10M) of dl-Nallylcamphoramic acid in 65 ml of methanol over a period of 30 minutes. The temperature of the reaction mixture should not rise over 30°C. The stirring is continued for one hour. The reaction mixture is allowed to stand at room temperature overnight in the dark to complete the reaction. A solution of 5.9 g (= 0.10M) of sodium chloride in 25 ml of water is added and the stirring is continued for four hours. The small amount of gray precipitate produced is removed by centrifuging. The colorless, clear supernatant is concentrated to about half of its original volume and then dropped into 300 ml of water with stirring.
The white precipitate which forms is filtered and dried at 80°C, yielding 45 g of chloromercuri acid (= 89% of the theory), MP 106° to 109°C (decomp.). This compound is finally obtained in analytically pure form and with a constant melting point by two recrystallizations from acetone-water giving a MP of 131° to 132°C with decomposition.
(B) Preparation of the Chloromercuri Acid Sodium Salt Solution: 50.6 g (= 0.100 M) of the chloromercuri acid (dried over CaCl2, at 0.1 mm and room temperature overnight) is dissolved in 100 ml of warm methanol. To this solution 6.0 g (= 0.111 M) of sodium methylate is added in small portions with constant stirring, so that the temperature of the solution does not rise over 30°C. The solution is centrifuged, and the glass is rinsed with 10 ml of methanol. The final pH of the combined solutions is 8.5.
(C) Preparation of the Disodium Thioglycolate Solution: The following steps are carried out under nitrogen. To 9.2 g (= 0.100 M) of freshly distilled thioglycolic acid (BP at 2 mm, 84° to 85°C) in 100 ml of methanol in a flask is added 12.0 g (= 0.222 M) of sodium methylate in small portions with stirring. The turbid solution is poured into a dropping funnel and the flask is rinsed with 20 ml of methanol. The final pH of the combined methanolic solutions is 11, according to US Patent 2,834,795.
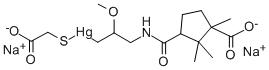
To 50 cc of a carefully purified aqueous solution of the sodium salt of N(γchloromercuri-β-methoxy-propyl)-d-α-camphoramic acid containing 40 mg of mercury per cc is added 10 cc of a solution containing 1.14 g (1 mol
equivalent) of sodium thioglycolate and the mixture is then evaporated to
dryness at room temperature and reduced pressure in the presence of a
desiccant. The product is an amorphous white powder which decomposes at
156° to 158°C (uncorr.), and which was found on analysis to have a mercury
content of 33.0%, according to US Patent 2,576,349.
Safety Profile
Poison by
subcutaneous and intravenous routes. When
heated to decomposition it emits very toxic
fumes of Hg, NOx, Na2O, and SOx. See also
MERCURY COMPOUNDS and
MERCAPTANS.