Synthesis
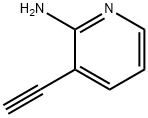
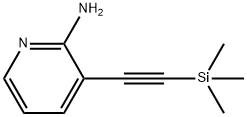
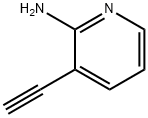
GENERAL STEPS: Preparation of 3-ethynylpyridin-2-amine; Tetrabutylammonium fluoride (1 M tetrahydrofuran solution, 20 mL, 20 mmol) was added to a tetrahydrofuran (300 mL) solution of 3-((trimethylsilyl)ethynyl)pyridin-2-amine (28.1 g, 148 mmol), and the reaction was stirred at room temperature for 15 minutes. Upon completion of the reaction, water was added to the reaction solution and then extracted four times with ethyl acetate. The organic layers were combined, dried with anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The residue was purified by silica gel column chromatography (eluent: heptane/ethyl acetate = 1:1, subsequently adjusted to 1:2) to afford the target product 3-ethynylpyridin-2-amine (16.4 g, 93.7% yield).1H-NMR (DMSO-d6) δ (ppm): 4.43 (1H, s), 6.14 (2H, brs), 6.53 (1H, dd, J =4.8 Hz, 7.2 Hz), 7.53 (1H, d, J=7.2 Hz), 7.96 (1H, d, J=4.8 Hz).
References
[1] Patent: US2009/82403, 2009, A1. Location in patent: Page/Page column 51-52
[2] Patent: US2007/105904, 2007, A1. Location in patent: Page/Page column 53-54
[3] Patent: US2010/331282, 2010, A1. Location in patent: Page/Page column 11
[4] Chemistry - A European Journal, 2010, vol. 16, # 27, p. 7992 - 7995
[5] Patent: EP2065377, 2009, A1. Location in patent: Page/Page column 59