Synthesis
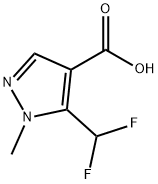
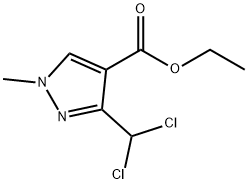
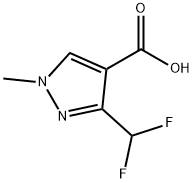
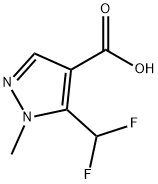
The general procedure for the synthesis of 3-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid and 5-(difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid using ethyl 3-(dichloromethyl)-1-methyl-1H-pyrazole-4-carboxylate as a raw material was as follows: according to the method of Example 13, 200 g of 3-(dichloromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid ethyl ester (GC purity: 92.8%, 0.783 mol) was added to an autoclave with 69.50 g of 1,3-dimethyl-2-imidazolidinone hexadecyl hydrofluoric acid salt (0.160 mol). The temperature was gradually increased to 130°C to 155°C. When the system temperature reached 130 to 155°C, the reaction was maintained at this temperature range for 7 to 12 hours. Upon completion of the reaction, the pressure was released, purged with nitrogen, and the exhaust gas was treated by alkaline absorption. The reaction mixture was cooled to 60 to 80°C and the reaction solution was diluted by adding 350 g of xylene and 60 g of water. Subsequently, 125.00 g of aqueous sodium hydroxide solution with a mass concentration of 30% (mass concentration refers to the percentage of sodium hydroxide to the total mass of the solution) was slowly added dropwise and stirred at 70°C for 1 hour. The mixture was separated and the aqueous phase was acidified with concentrated hydrochloric acid at a mass concentration of 36% (mass concentration refers to the percentage of hydrogen chloride to the total mass of the concentrated hydrochloric acid) to pH=1.0. Cooled to 10-25°C, filtered and dried under vacuum to give 117.60 g of 3-(difluoromethyl)-1 -methyl-1H-pyrazole-4-carboxylic acid (Yield: 84.5%, HPLC purity: 99.1%; isomer 5 -(Difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid with HPLC purity of 0.05%).
References
[1] Patent: CN106467492, 2017, A. Location in patent: Paragraph 0224; 0225; 0226; 0227; 0228; 0229; 0230-0235