Synthese
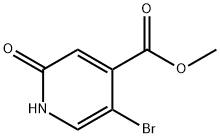
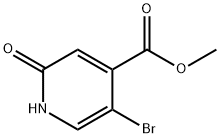
Step A: Synthesis of methyl 5-bromo-2-oxo-1,2-dihydropyridine-4-carboxylate-II
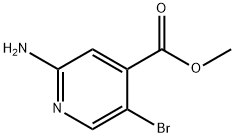
To a stirred solution of concentrated H2SO4 (50 mL, 0.668 mol) in water (500 mL) was added methyl 2-amino-5-bromoisonicotinate (I, 50 g, 0.226 mol). The resulting clarified brown solution was cooled to 0°C. A solution of NaNO2 (50 g, 0.668 mol) in water (150 mL) was added slowly and dropwise to the mixture at 0 °C using an addition funnel. During the reaction, vigorous bubble production was observed, accompanied by the escape of N2 gas. The reaction mixture was gradually warmed to room temperature and stirring was continued at room temperature for 30 minutes. Upon completion of the reaction, the solid product was collected by filtration and washed sequentially with water (3 x 150 mL) and hexane (2 x 100 mL) to give a yellow solid product (48.0 g, 93% yield).
Product characterization data:
1H NMR (DMSO-d6, 400 MHz), δ (ppm): 7.62 (s, 1H), 6.90 (s, 1H), 5.0 (br, 1H), 3.98 (s, 3H).
MS (ESI): mass calculated value C7H7BrN2O2, 232.01; m/z found, 234 [M + 2H]+.