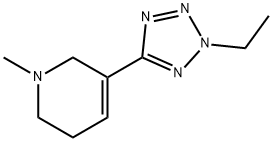
ALVAMELINE
- CAS号:120241-31-8
- 英文名:ALVAMELINE
- 中文名:ALVAMELINE
- CBNumber:CB92451399
- 分子式:C9H15N5
- 分子量:193.25
- MOL File:120241-31-8.mol
- 沸点 :342.1±52.0 °C(Predicted)
- 密度 :1.25±0.1 g/cm3(Predicted)
- 储存条件 :2-8°C
- 溶解度 :Soluble in DMSO
- 酸度系数(pKa) :7.29±0.40(Predicted)
ALVAMELINE性质、用途与生产工艺
- 生物活性 Alvameline (Lu25-109)是M1的部分激动剂,也是M2/M3拮抗剂。
-
体外研究
Alvameline is metabolized by human liver microsomes to Lu 31-126 mainly by CYP2D6; to Lu 29-297 and Lu 25-077 mainly by CYP1A2, CYP2A6, CYP2C19, and CYP3A4; and to Lu 32-181 by CYP1A2 and possibly by CYP2C19. One metabolite, Lu 32-181, could be reduced back to alvameline, a reaction not inhibited by the applied cytochrome P-450 inhibitors.
-
体内研究
Alvameline competitively and effectively antagonizes carbachol-induced contractions and contractions induced by electrical field stimulation in human detrusor muscle. Alvameline produces a concentration-dependent rightward shift of the concentration-response curves for carbachol in both human and pig detrusor, the pK b values being 6.2 and 5.8. Contractions induced by electrical field stimulation in human detrusor are almost completely inhibited by 100 μM alvameline. In contrast, electrical field stimulation-induced contractions in pig detrusor are less sensitive to alvameline, resulting in a final inhibition of 32% with the highest concentration used (100 μM). Alvameline has been shown to improve cognitive function following traumatic brain injury in rats. Alvameline treated rats causes a 13% and 5% decrease in the medial septal nucleus, a 48 and 23% decrease in the vertical limb nucleus of the diagonal band, and a 51 and 28% decrease in the nucleus basalis magnocellularis, respectively.
- 公司名称:DALTON
- 联系电话:--
- 电子邮件:chemist@dalton.com
- 国家:加拿大
- 产品数:966
- 优势度:78