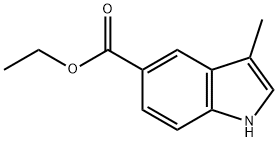
(1) Synthesis of 5-ethoxy-3-methylindole: To a solution of 2-bromo-4-ethoxy-N-allyl aniline (650 mg, 2.29 mmol) in acetonitrile (10 mL) was added sequentially tetrakis(triphenylphosphine)palladium(0) (132 mg, 0.114 mmol), palladium(II) acetate (25.7 mg, 0.114 mmol) and triethylamine (0.413 mL, 2.98 mmol). The reaction mixture was stirred in a sealed tube at 100 °C overnight. After completion of the reaction, the reaction mixture was filtered and the filtrate was concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (eluent: ethyl acetate/hexane = 1:2) to afford ethyl 3-methyl-1H-indole-5-carboxylate (340 mg, 73% yield). NMR hydrogen spectrum (CDCl3, method a): δ 1.43 (3H, t, J = 7.2 Hz), 2.37 (3H, s), 4.41 (2H, q, J = 7.2 Hz), 7.03 (1H, s), 7.34 (1H, d, J = 8.6 Hz), 7.91 (1H, dd, J = 1.3, 8.6 Hz), 8.08 (1H brs), 8.36 (1H, s).