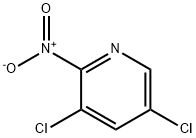
a) Synthesis of 3,5-dichloro-2-nitropyridine: 2-Amino-3,5-dichloropyridine (193 mg, 1.00 mmol) was dissolved in concentrated sulfuric acid (5 mL) and potassium persulfate (K2S2O8, 1.3 g, 5.0 mmol) was added batchwise. The reaction mixture was stirred at room temperature overnight and subsequently poured into crushed ice and neutralized with aqueous sodium bicarbonate (NaHCO3). The product was extracted with dichloromethane (CH2Cl2, 3 x 20 mL), and the organic phase was dried with anhydrous sodium sulfate (Na2SO4) and concentrated under reduced pressure to give the title compound (123 mg, 63.7% yield).1H-NMR (CDCl3, 400 MHz): δ 8.40 (d, 1H, J = 2.1 Hz), 8.05 (d, 1H, J = 2.1 Hz).