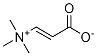
Uses
Reaction of allylic alcohols with (E)-(carboxyvinyl)trimethylammonium betaine affords (E)-3-(allyloxy)acrylic acids, which on heating are transformed to γ,δ-unsaturated aldehydes via Claisen rearrangement/decarboxylation.
Preparation
Preparative Methods of (E)-(Carboxyvinyl)trimethylammonium Betaine: A mixture of 25.0 g of ethyl propiolate, 14 mL of dichloromethane, and 440 mL of water
is cooled to 5 °C and 90 mL of an aqueous 25% solution of trimethylamine is added with vigorous stirring over a
period of 30 min. The mixture is then warmed to 25 °C for 3 h. The aqueous layer is separated and washed three
times with 100 mL of dichloromethane. The aqueous solution is concentrated in vacuo at 45 °C. When the
residue gives the appearance of a wet solid, it is treated with 150 mL of dioxane and concentrated as described
above. The dioxane treatment-concentration procedure is repeated three times. The resulting yellow solid is
triturated with acetonitrile until a white solid is obtained (25 g, 76% yield).
storage
(E)-(Carboxyvinyl)trimethylammonium Betaine will slowly decompose at room temperature over a period of
several months. Storage in a closed container at low temperatures is recommended. This reagent should be
handled in a fume hood.
Purification Methods
Purification of (E)-(Carboxyvinyl)trimethylammonium Betaine: yellow impurities can be removed by trituration with acetonitrile to obtain a white solid. This is
dried at 25 °C in vacuo (0.1 mmHg) for 14 h.