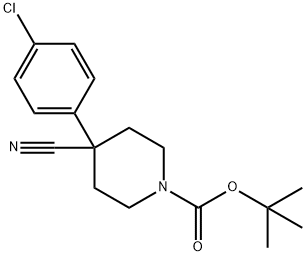
Step 1. 2-(4-Chlorophenyl)acetonitrile (20.1 g, 132.59 mmol, 1.00 eq.) and tert-butyl N,N-bis(2-chloroethyl)carbamate (35.4 g, 146.19 mmol, 1.10 eq.) were dissolved in anhydrous DMF (200 mL) protected by nitrogen and cooled to 0°C. The mixture was then dissolved in a mixture of sodium hydroxide (27 g, 60% mineral oil dispersion, 666.73 mmol, 3.00 eq.) and sodium hydride (27 g). Sodium hydride (27 g, 60% mineral oil dispersion, 666.73 mmol, 3.00 eq.) was added in batches over 2 hours. The reaction mixture was stirred at 60 °C for 1.5 h, followed by stirring at room temperature overnight. Upon completion of the reaction, the reaction was quenched by careful addition of 250 mL of saturated aqueous NaHCO3. The reaction mixture was extracted with dichloromethane (3 x 200 mL), the organic phases were combined, washed with brine (2 x 300 mL), dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography with the eluent dichloromethane/petroleum ether (1:1) to afford tert-butyl 4-(4-chlorophenyl)-4-cyanopiperidine-1-carboxylate (30 g, 71%) as a yellow solid.TLC: Rf = 0.15 (Expanding agent: ethyl acetate/petroleum ether=1/5).