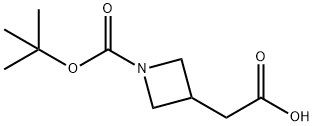
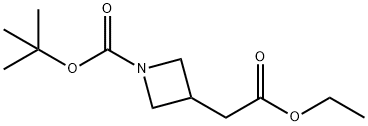
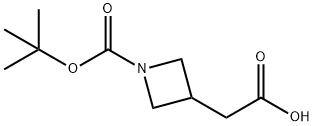
General procedure for the synthesis of N-Boc-3-azetidineacetic acid from tert-butyl 3-(2-ethoxy-2-oxoethyl)azetidine-1-carboxylate: lithium hydroxide (862 mg, 36.0 mmol) was added to tert-butyl 3-(2-ethoxy-2-oxoethyl)azetidine-1-carboxylate (7.30 g, 30.0 mmol) in a tetrahydrofuran ( 40 mL) and water (10 mL) in a mixed solution of tetrahydrofuran (40 mL) and water (10 mL) and the reaction mixture was stirred at 20 °C for 2 hours. The pH of the reaction mixture was adjusted to 4 by addition of 1 M aqueous hydrochloric acid, followed by extraction with ethyl acetate (3 x 100 mL). The organic layers were combined, washed sequentially with water (100 mL) and saturated aqueous sodium chloride (100 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to afford N-Boc-3-azetidineacetic acid as a white solid. Yield: 6.0 g, 28 mmol, 93% yield.1H NMR (400 MHz, CD3OD) δ 4.06 (dd, J = 8.5, 8.3 Hz, 2H), 3.61 (dd, J = 8, 6 Hz, 2H), 2.94-2.82 (m, 1H), 2.62 (d, J = 7.8 Hz, 2H), 1.43 (s, 9H) .