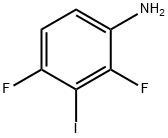
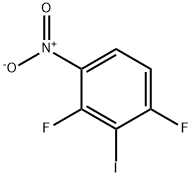
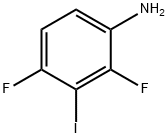
General procedure for the synthesis of 2,4-difluoro-3-iodoaniline from 1,3-difluoro-2-iodo-4-nitrobenzene: To a solution of concentrated hydrochloric acid (4 mL) of 1,3-difluoro-2-iodo-4-nitrobenzene (1.14 g, 4 mmol) was added anhydrous stannous chloride (2.71 g, 12 mmol), and the reaction mixture was heated to 50°C and kept for 1 hour. After completion of the reaction, the mixture was diluted with water, neutralized with 5% aqueous sodium hydroxide solution to pH=9, and then extracted with dichloromethane. The organic phase was washed with saturated saline, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to afford the target product 2,4-difluoro-3-iodoaniline (0.93 g, 91% yield).1H NMR (CDCl3): δ 6.70-6.93 (2H, m), 6.65 (2H, br).