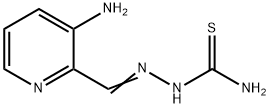
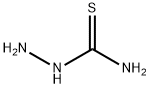
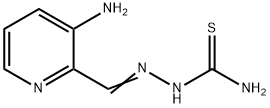
General procedure for the synthesis of [(3-aminopyridin-2-yl)methylideneamino]thiourea from amino thiourea and 2-(1,3-dioxolan-2-yl)-3-aminopyridine (CAS:143621-33-4):
Example VII: To a solution of 2-(1,3-dioxolan-2-yl)-3-aminopyridine (0.80 g, 4.8 mmol) dissolved in 10 mL of ethanol, 8 mL of water, and 2 mL of concentrated hydrochloric acid was added aminothiourea (0.48 g, 5.3 mmol). The reaction mixture was stirred at room temperature overnight followed by refluxing for 1 hour. It was cooled and filtered to give crude yellow hydrochloride. The hydrochloride was dissolved in 50 mL of hot water and filtered. To the hot filtrate was added 10 mL of 5% sodium bicarbonate solution. The mixture was stirred at room temperature for 1 h. The mixture was filtered and washed sequentially with water and ethanol to give 3-amino-2-formylpyridine acetylthiourea. Yield: 0.72 g (77%); melting point: 240-241°C. Mass spectrum (MS) m/e 194 (M+); 1H NMR (90 MHz, DMSO-d6) δ 6.48 (br s, 2H, 3-NH2, exchangeable in D2O), 7.12 (m, 2H, 4-H and 6-H), 7.83 (dd, 1H, 5-H). 8.10 (br s, 2H, CSNH2, exchangeable for D2O), 8.10 (s, 1H, 2-CH), 10.95 (s, 1H, NNH, exchangeable for D2O). Elemental analysis (C7H9N5S) is consistent with the theoretical values of C, H and N.