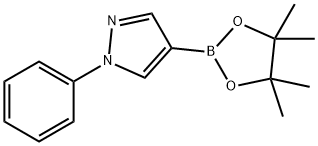
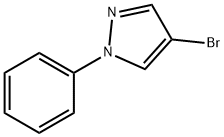
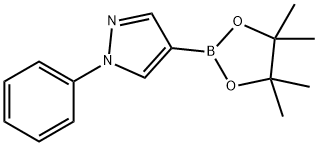
The general procedure for the synthesis of 1-phenyl-1H-pyrazole-4-boronic acid pinacol ester from 4-bromo-1-phenyl-1H-pyrazole and biboronic acid pinacol ester was as follows: to a solution of 4-bromo-1-phenyl-1H-pyrazole (0.5 g, 2.3 mmol) in DMSO (50 mL) was added sequentially at room temperature KOAc (0.66 g, 6.8 mmol), bis( pinacolato) diboron (0.63 g, 2.5 mmol) and PdCl2(dppf) (0.076 g, 0.11 mmol). The reaction mixture was stirred at 80 °C overnight. After completion of the reaction, the mixture was diluted with ethyl acetate, washed with saturated brine, dried over anhydrous sodium sulfate, concentrated and purified by column chromatography (eluent ratio: ethyl acetate:petroleum ether = 1:12) to afford 1-phenyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (0.3 g, 50% yield) as a light yellow solid.