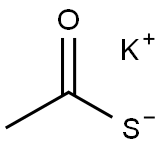
-
外観
白色~褐色, 結晶性粉末
-
溶解性
水及びエタノールにやや溶けにくく、アセトンにほとんど溶けない。
-
用途
有機合成原料、重金属沈殿剤。
-
使用上の注意
不活性ガス封入。
-
化学的特性
white to light brown crystalline powder, crystals
-
使用
Potassium Thioacetate is the potassium salt of Thioacetic Acid, a commonly used reagent in organic synthesis for the introduction of thiol groups in molecules.
-
主な応用
Potassium thioacetate is an organosulfur compound and a salt with the formula CH3COS−K+. This white, water-soluble solid is used as a reagent for preparing thioacetate esters and other derivatives . It acts as a sulfur source in the synthesis of sulfur-containing organic compounds for the synthesis of heterocycles, polymers, transition-metal ligands, nanoparticles, bioactive compounds and macromolecular inclusion complexes. It is also used for palladium mediated coupling with aryl halides and triflates leading to S-arylthioacetates and derivatives and it is also used as a reagent in the conversion of halides to thiols.
-
反応性
Thioacetate is also a class of sulfur-containing nucleophiles, of which potassium thioacetate is the most widely used reagent. Potassium thioacetate reacts with organic halides to form thioesters, which are often used as thiol-protecting groups.In the classical reaction, potassium thioacetate replaces the bromine atom to form a thiol in which the thioester formed in the first step is subjected to a nucleophilic addition elimination reaction to obtain a thiol via hydrolysis, alcoholysis or ammonolysis. Unlike the thiol formation from bromine and thiourea, this method is not strictly limited to polystyrenes. It can also be extended to poly(meth)acrylate systems. It is important that there is no observation of ester hydrolysis during the process.