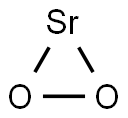
-
用途
医薬品、花火、曳光弾
-
化学的特性
Strontium peroxide is a lightcolored solid of good thermal stability. Commercial strontium peroxide contains about 85% SrO2 and 10% active oxygen.
-
使用
Strontium peroxide or strontium dioxide (SrO2) can cause fires or explode when heated and in contact with organic substances. It is used as both a reducing agent and an oxidizing agent.
-
製造方法
Like the other alkaline earth peroxides, it can be
prepared by reaction of the nitrate and sodium peroxide
in a cold solution:
Sr(NO3)2+ Na2O2+xH2O
SrO2·xH2O+2NaNO3
The hydrated form is usually the octahydrate. If the
anhydrate is desired, the hydrated peroxide is dried
and then sintered at 350°C for 10 min or less:
SrO2·xH2O+ heat→SrO+SrO2+H2O
-
一般的な説明
A white powder. Insoluble in water and slowly decomposed by water. Noncombustible, but accelerates the burning of combustible material.
-
空気と水の反応
Insoluble in water and slowly decomposed by water.
-
反応プロフィール
Strontium peroxide may explode from friction, heat or contamination. Accelerates burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). If the combustible material is finely divided the mixture may be explosive.
-
健康ハザード
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution.
-
火災危険
May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard.
-
安全性プロファイル
A powerful oxidizer. A skin, eye, and mucous membrane irritant. Mixtures with organic materials readily ignite with friction or on contact with moisture. See also PEROXIDES and STRONTIUM COMPOUNDS.