酵素阻害剤
This orally active, non-ATP-competitive cyclin kinase-directed inhibitor
(FW = 483.99 g/mol (mono-HCl); CASs = 827022-32-2 (mono-
hydrochloride, 571190-30-2 (free base); Solubility: 10 mg/mL DMSO; 30
mg/mL Water; Formulation: Dissolved in sodium lactate buffer (50 mM,
®
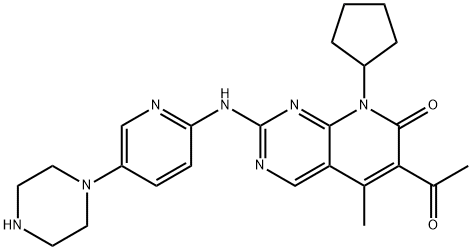
pH 4.0) ), also known as PD-0332991, Ibrance, and 6-acetyl-8-cyclopentyl-
5-methyl-2- (5- (piperazin-1-yl) pyridin-2-ylamino) pyrido[2,3-d]pyrimidin-
7 (8H) -one hydrochloride, targets Cdk-4 (Cyclin D1) and Cdk-6 (Cyclin D2),
enzymes that participate in the so-called CDK4/6-retinoblastoma signaling
pathway governing the cell-cycle restriction point. Palbociclib induces rapid
G1 cell-cycle arrest in primary human myeloma cells. This agent also
shows significant efficacy in a broad spectrum of human tumor xenografts
in vivo, resulting in complete regression in some tumors with no evidence of
acquired resistance or ability to circumvent the growth inhibitory properties
of this agent. Ibrance received FDA approval in 2015 for combined
use with letrozole to treat postmenopausal women with estrogen receptor-
positive, (HER2) -negative advanced breast cancer as an initial endocrine-
based therapy for metastatic disease. Cyclin Target Selectivity: Cdk1 (weak,
if any), Cdk2 (weak, if any), Cdk3 (weak, if any), Cdk4 (IC50 = 11 nM),
Cdk5 (weak, if any), Cdk6 (IC50 = 16 nM), Cdk7 (weak, if any), Cdk8
(weak, if any), Cdk9 (weak, if any), Cdk10 (weak, if any).