-
外観
白色~ほとんど白色, 結晶性粉末
-
定義
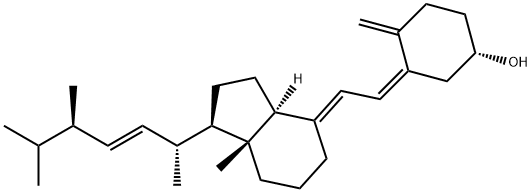
本品は、ビタミンD2であり、次の化学式で表される。
-
溶解性
水に殆ど不溶。エタノール, アセトンに易溶。エタノール及びアセトンに溶けやすく、水にほとんど溶けない。
-
解説
ビタミン D2:C28H44O(396.65).エルゴカルシフェロール(ergocalciferol)ともいう.無色の針状または柱状結晶.融点115~118 ℃.λmax 265 nm.[α]20D+103~+106°(エタノール).ビタミン D3 とともに多くの有機溶媒,油脂に可溶,水に不溶.エルゴステロールの紫外線照射(最適波長275~300 nm)によってつくられる.ビタミン D2,D3 はともに空気中で不安定であるが,窒素中-20 ℃ で保存すれば比較的安定である.ビタミン D2 の抗くる病作用は白ネズミ予防試験では40000国際単位/1 mg で,ビタミン D3 と同等である.ビタミンDの活性単位はエルゴカルシフェロール0.025 μg のもつ生物学的活性と規定されている.
-
用途
ビタミン D2 化合物です。
-
用途
薬理研究用
-
化粧品の成分用途
皮膚コンディショニング剤
-
効能
くる病治療薬, ビタミンD受容体作動薬
-
説明
Vitamin D aids in the absorption of calcium and has central roles in bone formation and maintenance, hypertension, cancer and immunity. Vitamin D may be obtained from many dietary sources, including eggs and fish, and is synthesized in the skin by the conversion of 7-dehydrocholesterol to vitamin D3 by ultraviolet light. Vitamin D2 is produced in fungi, including yeast, and invertebrates from ergosterol in response to ultraviolet radiation. In vertebrates as well as host organisms, vitamin D2 is metabolized first to 25-hydroxyvitamin D2 and subsequently to the active 1,25-dihydroxyvitamin D2. Differences in the metabolism and action of vitamin D2 vs. vitamin D3 in mammals is a current topic of research interest.
-
化学的特性
Ergocalciferol is an odorless white crystalline
solid
-
使用
Vitamin d2 is fat-soluble, and is stable unless oxidized. It is necessary for growth and maintenance of teeth and bones and the normal utilization of calcium and phosphorus; it is used medicinally in the treatment of rickets and as a dietary supplement. Its sources include fish liver and vitamin d-fortified milk.
-
定義
ChEBI: A vitamin D supplement and has been isolated from alfalfa.
-
一般的な説明
Odorless white crystals. Used as a dietary supplement and food additive.
-
反応プロフィール
Flammable and/or toxic gases are generated by the combination of alcohols with alkali metals, nitrides, and strong reducing agents. They react with oxoacids and carboxylic acids to form esters plus water. Oxidizing agents convert them to aldehydes or ketones. Alcohols exhibit both weak acid and weak base behavior. They may initiate the polymerization of isocyanates and epoxides.
-
健康ハザード
Vitamin D2 poisoning disturbs calcium metabolism and causes kidney damage. Vitamin D2 in a single acute ingestion presents no toxic hazards. Daily ingestion in excess of 5000 units/day in children or 7500 units/day in adults will produce toxic symptoms associated with hypervitaminosis D. Those with hypercalcemia are at a greater risk.
-
火災危険
Shows signs of decomposition when stored for a few days at room temperature.
-
安全性プロファイル
Poison by ingestion,
intraperitoneal, intravenous, and
intramuscular routes. An experimental
teratogen. Human systemic effects by
ingestion: anorexia, nausea or vomiting, and
weight loss. Experimental reproductive
effects. When heated to decomposition it
emits acrid smoke and irritating fumes.
-
職業ばく露
Used as a nutrient and/or dietary supplement food additive
-
代謝経路
Ergocalciferol (vitamin D2) is a product made by the UV irradiation of
yeast ergosterol. It is a common form of vitamin D and it has very similar
biological activity to that of cholecalciferol (vitamin D3) in the treatment of
the vitamin D deficiency diseases (e.g. rickets). The bioactive form of this
vitamin is also the 1,25-dihydroxy-derivative and this has equal antirachitic
activity to that of the cholecalciferol analogue (Jones et al., 1975).
-
輸送方法
UN2811 Toxic solids, organic, n.o.s., Hazard
Class: 6.1; Labels: 6.1-Poisonous materials, Technical
Name Required. UN3249 Medicine, solid, toxic, n.o.s.,
Hazard Class: 6.1; Labels: 6.1-Poisonous materials
-
純化方法
It is converted into its 3,5-dinitrobenzoyl ester and crystallised repeatedly from acetone. The ester is then saponified and the free vitamin is isolated. [Laughland & Phillips Anal Chem 28 817 1956, Beilstein 6 IV 4404.]
-
不和合性
Dust may be combustible and may form
explosive mixture with air. Incompatible with oxidizers
(chlorates, nitrates, peroxides, permanganates, perchlorates,
chlorine, bromine, fluorine, etc.); contact may cause fires
or explosions. Keep away from alkaline materials, strong
bases, strong acids, oxoacids, and epoxides.
-
廃棄物の処理
Dispose of contents and container to an approved waste disposal plant. All federal,
state, and local environmental regulations must be
observed. It is inappropriate and possibly dangerous to the
environment to dispose of expired or waste drugs and pharmaceuticals by flushing them down the toilet or discarding
them to the trash. Household quantities of expired or waste
pharmaceuticals may be mixed with wet cat litter or coffeegrounds, double-bagged in plastic, discard in trash. Larger
quantities shall carefully take into consideration applicable
DEA, EPA, and FDA regulations. If possible return the
pharmaceutical to the manufacturer for proper disposal
being careful to properly label and securely package the
material. Alternatively, the waste pharmaceutical shall be
labeled, securely packaged and transported by a state
licensed medical waste contractor to dispose by burial in a
licensed hazardous or toxic waste landfill or incinerator.