説明
Similar to omarigliptin,
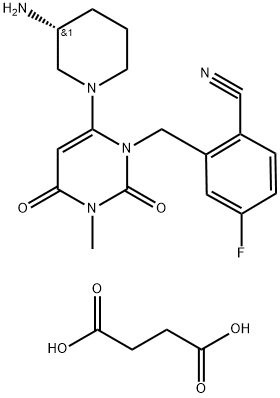
trelagliptin succinate (XIX) is a highly selective, orally
delivered inhibitor of DPP-4 developed by Takeda Pharmaceuticals
and approved in Japan in March 2015 for the
treatment of type 2 DM. Interestingly, trelagliptin is
structurally similar to alogliptin, a DPP-4 inhibitor also
marketed by Takeda and described in our 2010 review,
differing only in the presence of a fluorine in the 5-position of
the cyanobenzyl moiety. Both trelagliptin and alogliptin are
potent inhibitors of DPP-4, with IC50s of 1.3 and 5.3 nM,
respectively. Notably, while similar drugs are dosed once
daily, trelagliptin is the first DPP-4 inhibitor approved for onceweekly
dosing. Kinetic analysis has revealed that trelagliptin is a
substrate-competitive, reversible, slow-binding inhibitor (t1/2 for
dissociation = ca. 30 min) of DPP-4, although the dissociation
time is insufficient to explain its long-acting effects. In a phase
III trial, once-weekly trelagliptin (100 mg) showed similar
efficacy and safety to once-daily alogliptin (25 mg) in patients
with type 2 DM inadequately controlled by diet and exercise.
The medicinal chemistry discovery of trelagliptin and
alogliptin as well as reviews of this class of compounds
have been published.