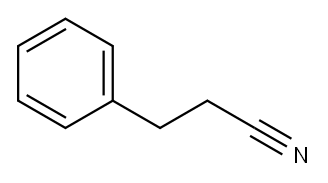
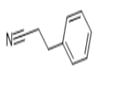
clear colorless to light yellow liquid
3-Phenylpropionitrile was used to study the free enzyme activity of nitrilase AtNIT1.
A glucosinolate enzymic hydrolosis product with potential antibacterial activities against plant pathogenic bacteria
To a stirred solution of 3-phenylpropanamide (50 mg, 0.34 mmol) in acetonitrile (0.8 mL) at room temperature were successively added formic acid (0.2 mL) and paraformaldehyde (50 mg, 1.67 mmol). The reaction mixture was then refluxed for 12 h, and the solution obtained was cooled to room temperature.
Work-up A: The crude mixture was concentrated under reduced pressure, and the residue was subjected to flash chromatography on silica gel (230–240 mesh) eluting with hexane/ethyl acetate (7:3) to yield 3-phenylpropanonitrile (37 mg, 85%).
Work-up B: The reaction mixture was diluted with ethyl acetate (10 mL) and washed successively with saturated sodium hydrogen carbonate solution (5 mL) and water (5 mL). The combined aqueous layers were extracted with ethyl acetate (2 × 10 mL). The combined organic layers were dried over Na2SO4, filtered, and concentrated. The crude 3-phenylpropanonitrile was purified as described above (Work-up A).
ChEBI: A nitrile that is propionitrile in which one of the methyl hydrogens has been replaced by a phenyl group.
The effect of 3-Phenylpropionitrile on the growth and survival of woodlouse Porcellio scaber has been studied.