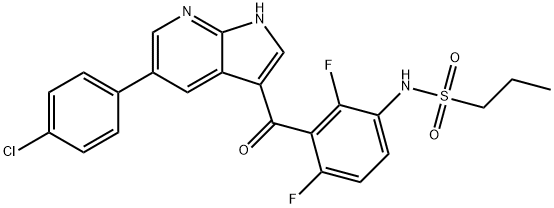
-
効能
抗悪性腫瘍薬, BRAFキナーゼ阻害薬
-
商品名
ゼルボラフ (中外製薬)
-
説明
In August 2011, the United States FDA approved vemurafenib (PLX-
4032, RO-5185426) for the treatment of patients with metastatic melanoma
with the BRAFV600E mutation. Vemurafenib has been developed as a
targeted therapy for patients with the BRAF gene mutation since oncogenic
B-raf signaling is implicated in approximately 50% of melanomas. Vemurafenib was identified based on an initial high-throughput screen followed by the extensive use of structure-based drug design. Vemurafenib is a potent inhibitor of B-RafV600E kinase (IC50=13 nM) compared to its potency against wildtype B-raf (IC50=160 nM) and is fairly selective versus a panel of 200
kinases. It does inhibit other kinases (RAF1, SRMS, ACK1,
MAP4K5, and FGR) and mutant B-raf kinases (BRAFV600K,
BRAFV600D, and BRAFV600R) with enzyme IC50's of <100 nM.
-
化学的特性
Off-White Solid
-
使用
Vemurafenib selective BRAFV600E kinase inhibitor; an antitumor agent. Vemurafenib functions by inhibiting the proliferation and mitogen-activated protein/extracellular signal-regulated kinase (ERK) kinase and ERK phosphorylation in a panel of tumor cell lines, including melanoma cell lines expressing BRAFV600E or other mutant BRAF proteins altered at codon 600.
-
定義
ChEBI: A pyrrolopyridine that is 1H-pyrrolo[2,3-b]pyridine which is substituted at position 5 by a p-chlorophenyl group and at positions 3 by a 3-amino-2,6-difluorobenzoyl group, the amino group of which has undergone
ormal condensation with propane-1-sulfonic acid to give the corresponding sulfonamide. An inhibitor of BRAF and other kinases.