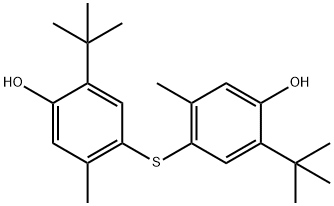
Chemical Properties
4,40-Thiobis(6-tert-butyl-m-cresol) is a light gray to tan powder. Aromatic odor.
Chemical Properties
white to almost white crystals or crystalline
Uses
Protection of light-colored rubber from oxidation
and of nonstaining neoprene compounds
against deterioration.
Uses
4,4''-Thiobis(2-tert-butyl-5-methylphenol) is an antioxidant. 4,4''-Thiobis(2-tert-butyl-5-methylphenol) is a radical inhibitor which prevented thermal decomposition of the peracid.
Uses
As an antioxidant in the rubber and
plastics industry; as a stabilizer in polyethylene
and polyolefin packaging materials for foodstuffs.
General Description
White or light gray to tan powder.
Air & Water Reactions
Sensitive to base hydrolysis and may spontaneously oxidize in solution. . Insoluble in water.
Reactivity Profile
4,4'-Thiobis(6-tert-butyl-m-cresol) may be sensitive to light.
Hazard
Toxic by inhalation. Upper respiratory tract
irritant. Questionable carcinogen.
Health Hazard
4,4'-Thiobis(6-tert-butyl-mcresol)
(TBBC) is of low systemic toxicity in
animals; allergic contact dermatitis has been
reported in humans.
Fire Hazard
4,4'-Thiobis(6-tert-butyl-m-cresol) is combustible.
Flammability and Explosibility
Non flammable
Safety Profile
Poison by
intraperitoneal route and probably by
ingestion and inhalation. Mutation data
reported. See also SULFIDES. When heated
to decomposition it emits highly toxic fumes
of SOx.
Potential Exposure
This material is used as an antioxidant in the plastics and rubber industries; in Neoprene and other synthetic rubbers; in polyethylene and polypropylene.
Carcinogenicity
TBBC was not mutagenic in Salmonella
typhimurium strains with or without metabolic
activation. In Chinese hamster ovary cells,
TBBC induced an increase in sister chromatid
exchanges but there were no increases in chromosomal
aberrations.
Incompatibilities
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. May evolve explosive hydrogen sulfide upon contact with moisture or acids.