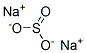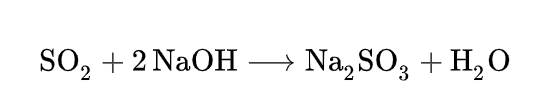Chemical Properties
Upon contact with strong or weak acids, Na2SO3 undergoes decomposition, liberating gaseous sulphur dioxide.
Sodium sulfite reacts with aldehydes to yield a bisulfite adduct. However, sulfonic acids are produced from their reaction with ketones.
Solutions of sodium sulfite are oxidised by atmospheric oxygen, yielding sodium sulphate.
This compound is insoluble in ammonia and chlorine.
Uses
sodium sulphite is a white, water-soluble solid and used commercially as an antioxidant and preservative. It is also suitable for the softening of lignin in the pulping and refining processes of wood and lignocellulosic materials.
Preparation
Sodium sulfite can be prepared by treating a solution of sodium hydroxide with sulfur dioxide. When conducted in warm water, Na2SO3 initially precipitates as a white solid. With more SO2, the solid dissolves to give the disulfite, which crystallizes upon cooling.
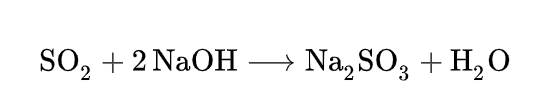
Structure and conformation
According to X-ray crystallography sodium sulfite heptahydrate features pyramidal SO32- centers. The S-O distances are 1.50 and the O-S-O angles are near 106º.
Toxicity evaluation
Sodium sulfite produces toxic and corrosive fumes. It is a strong reducing agent. It reacts violently with oxidants.