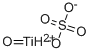General Description
A colorless or violet colored liquid. Dilution releases heat and may cause precipitation of solid titanium sulfate. Corrosive to metals and tissue. TITANIUM SULFATE, SOLUTION(13825-74-6) is used to make pigments and in dyeing.
Reactivity Profile
A highly acidic liquid.
Air & Water Reactions
Can generate heat on dilution with water.
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Chemical Properties
White, cake-like solid. Highly acidic,
similar to 50% sulfuric acid, typical composition
20% TiO2, 50% sulfuric acid, 30% water,
hygroscopic. Soluble in water; solutions
hydrolyze readily unless protected from heat and
dilution.
Uses
TiOSO4 (titanium (IV) oxysulfate) was used for the preparation of stable amorphous titanium oxide (TiO2) hollow microspheres.
Uses
In tanning; in production of pigment grade and of anatase-structured titanium dioxide.
Hazard
Irritant to skin and tissue.
Purification Methods
Dissolve it in water, filter and crystallise it three times from boiling 45% H2SO4, washing with EtOH to remove excess acid, then with Et2O. Dry it in air for several hours, then in an oven at 105-110o. [Hixson & Fredrickson Ind Eng Chem 37 678 1945.]