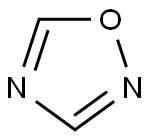
1,2,4-оксадиазол
- английское имя1,2,4-oxadiazole
- CAS №288-90-4
- CBNumberCB91564357
- ФормулаC2H2N2O
- мольный вес70.05
- файл Mol288-90-4.mol
химическое свойство
| Температура плавления | 69-70 °C(Solv: ethanol (64-17-5)) |
| Температура кипения | 87 °C |
| плотность | 1.193±0.06 g/cm3(Predicted) |
| пка | -0.28±0.20(Predicted) |
1,2,4-оксадиазол химические свойства, назначение, производство
Описание
1,2,4-Oxadiazole is a five-membered, planar, conjugated, nonbenzonoid, aromatic heterocyclic system comprised of one oxygen atom and two nitrogen atoms at positions 2 and 4 of the oxadiazole ring. The oxadiazole ring has two pyridine-like nitrogen atoms. Positions 3 and 5 are vulnerable for nucleophilic substitution to form 3,5-disubstituted 1,2,4-oxadiazoles. The topography of the 1,2,4-oxadiazole ring reveals that it has a more heterodiene character compared to its aromatic nature. The substituted 1,2,4-oxadiazole ring system was first reported in 1880 but the parent 1,2,4-oxadiazole was not reported until 1962 because of instability of the molecule.Физические свойства
The parent 1H-1,2,4-oxadiazole is highly unstable and its synthesis has not been reported so far. The 3,5-disubstituted 1,2,4-oxadiazoles are stable, while monosubstituted oxadiazoles are less stable and prone to acid- and base-induced hydrolysis. The 13C NMR spectra of 3,5-diaryl-1,2,4-oxadiazoles in CDCl3 showed chemical shift for C3 at 168.8–169.0 ppm and C5 at 174.7–175.8 ppm.Использование
1,2,4-Oxadiazole can be used in biological study of antitubercular activity of butyrate-specific oxadiazoles against Mycobacterium tuberculosis and their cytotoxicity on monkey kidney cells.