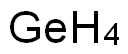
Германий
- английское имяGermanium
- CAS №7440-56-4
- CBNumberCB7733835
- ФормулаGe
- мольный вес72.64
- EINECS231-164-3
- номер MDLMFCD00085310
- файл Mol7440-56-4.mol
| Температура плавления | 937 °C (lit.) |
| Температура кипения | 2830 °C (lit.) |
| плотность | 5.35 g/mL at 25 °C (lit.) |
| давление пара | 0Pa at 25℃ |
| температура хранения | Flammables area |
| растворимость | insoluble in H2O, dilute acid solutions, alkaline solutions |
| форма | powder |
| цвет | Silver |
| Удельный вес | 5.35 |
| РН | 0.5 (20°C in H2O) |
| Flame Color | Pale blue |
| удельное сопротивление | 53000 μΩ-cm, 20°C |
| Растворимость в воде | insoluble H2O, HCl, dilute alkali hydroxides; attacked by aqua regia [MER06] |
| Кристальная структура | Cubic, Diamond Structure - Space Group Fd3m |
| Мерк | 13,4419 |
| Пределы воздействия | ACGIH: TWA 0.5 ppm(2.5 mg/m3); Ceiling 2 ppm (Skin) OSHA: TWA 3 ppm NIOSH: IDLH 30 ppm(250 mg/m3); TWA 3 ppm(2.5 mg/m3); Ceiling 6 ppm(5 mg/m3) |
| Стабильность | Stable. Slightly soluble in strong acids. Incompatible with strong oxidizing agents. |
| LogP | 4.14 at 20℃ |
| Справочник по базе данных CAS | 7440-56-4(CAS DataBase Reference) |
| FDA UNII | 00072J7XWS |
| Справочник по химии NIST | Germanium(7440-56-4) |
| Система регистрации веществ EPA | Germanium (7440-56-4) |
| UNSPSC Code | 41116107 |
| NACRES | NA.24 |
| Коды опасности | F,Xi | |||||||||
| Заявления о рисках | 36/37/38-36/38-11 | |||||||||
| Заявления о безопасности | 26-36/39-2 | |||||||||
| РИДАДР | UN 3089 4.1/PG 2 | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | LY5200000 | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 8 | |||||||||
| Группа упаковки | III | |||||||||
| кода HS | 28053090 | |||||||||
| Банк данных об опасных веществах | 7440-56-4(Hazardous Substances Data) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)



-
сигнальный язык
опасность
-
вредная бумага
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
H373:Может поражать органы (Нервная система) в результате многократного или продолжительного воздействия при вдыхании.
H361:Предполагается, что данное вещество может отрицательно повлиять на способность к деторождению или на неродившегося ребенка.
H228:Воспламеняющееся твердое вещество.
-
оператор предупредительных мер
P201:Беречь от тепла, горячих поверхностей, искр, открытого огня и других источников воспламенения. Не курить.
P202:Перед использованием ознакомиться с инструкциями по технике безопасности.
P210:Беречь от тепла, горячих поверхностей, искр, открытого огня и других источников воспламенения. Не курить.
P240:Заземлить и электрически соединить контейнер и приемное оборудование.
P273:Избегать попадания в окружающую среду.
P308+P313:ПРИ подозрении на возможность воздействия обратиться за медицинской помощью.
Германий химические свойства, назначение, производство
Описание
Germanium is extracted from zinc ores in a very complicated process as it has aqueous properties similar to those of zinc. Once the germanium/zinc mixture has been sufficiently enriched with germanium, it is heated in HCl with Cl2 in order to allow the formation of germanium tetrachloride (GeCl2).Химические свойства
Germanium is a grayish-white, lustrous, and brittle metalloid. The powder is grayish-black and odorless. It is never found free and occurs most commonly in ergyrodite and germanite. It is generally recovered as a by-product in zinc production, coal processing, or other sources.Физические свойства
Germanium has a gray shine with a metallic silvery-white luster. It is a brittle elementclassed as a semimetal or metalloid, meaning it is neither a metal such as iron or copper nora nonmetal, such as phosphorus, sulfur, or oxygen. Germanium has some properties likea metal and some like a nonmetal. It is a crystal in its pure state, somewhat like silicon. Itwill combine with oxygen to form germanium dioxide, which is similar to silicon dioxide(sand).Germanium is not found in its free elemental state because it is much too reactive. For themost part, it is found combined with oxygen, either as germanium monoxide or as germaniumdioxide. Also, it is recovered from the ores of zinc, copper, and arsenic and the flue depositsof burning coal.
The crystal structure of germanium is similar to that of diamonds and silicon, and its semiconductingproperties are also similar to silicon.The melting point of germanium is 938.3°C, its boiling point is 2833°C, and its densityis 5.323 g/cm3.
Изотопы
There are a total of 38 isotopes of Germanium, five of which are stable. Thestable isotopes of germanium and their natural abundance are as follows: Ge-70 =20.37%, Ge-72 = 27.31%, Ge-73 = 7.76%, Ge-74 = 36.73%, and Ge-76 = 7.83%.Ge-76 is considered stable because it has such a long half-life (0.8×10+25 years)All theother 33 isotopes are radioactive and are produced artificially.Происхождение имени
Germanium’s name was derived from the Latin word Germania, meaning “Germany.”Вхождение
Germanium, the 52nd most abundant element in the Earth’s crust, is widely distributed,but never found in its natural elemental state. It is always combined with other elements,particularly oxygen.Germanium’s main minerals are germanite, argyrodite, renierite and canfieldite, all ofwhich are rare. Small amounts of germanium are found in zinc ore, as well as in copper andarsenic ores. It is known to concentrate in certain plants on Earth, particularly in coal: commercialquantities are collected from the soot in the stacks where coal is burned.
Характеристики
Once germanium is recovered and formed into blocks, it is further refined by the manufacturerof semiconductors. It is melted, and the small amounts of impurities such as arsenic, gallium,or antimony, are added. They act as either electron donors or acceptors that are infused(doped) into the mix. Then small amounts of the molten material are removed and used togrow crystals of germanium that are formed into semiconducting transistors on a germaniumchip. The device can now carry variable amounts of electricity because it can act as both aninsulator and a conductor of electrons, which is the basis of modern computers.Использование
In electronics: manufacture of rectifying devices (germanium diodes), transistors, in red-fluorescing phosphors; in dental alloys; in the production of glass capable of transmitting infrared radiation. Review of uses: Aldington, Cumming, Endeavour 14, 200-204 (1955); New Uses for Germanium, F. I. Metz, Ed. (Midwest Research Institute, 1974) 120 pp.Методы производства
The concentration of germanium in the earth’s crust is approximately 7 ppm. Germanium is not found in the free state, but in combination with other elements as a mineral, such as in the mineral argyrodite (Ag8GeS6, 5–7% Ge) and germanite (7CuS–FeS2–GeS2, 8.7% Ge). Enargite, a Cu–As sulfide, is found in the western United States and contains as much as 0.03% Ge; however, none of these minerals are utilized for recovery of germanium because of the small quantities available. The principal domestic source of germanium is from the residues of cadmium derived from zinc ores. Commercial recovery of germanium has been chiefly from zinc and Zn–Cu–Pb ores, germanite, and flue dusts from coals. Some silver and tin ores contain germanium, as do many types of coal. Oak and beech humus in one locality in Germany reportedly contain 70 ppm germanium.Определение
A hard brittle gray metalloid element belonging to group 14 (formerly IVA) of the periodic table. It is found in sulfide ores such as argyrodite (4Ag2S·GeS2) and in zinc ores and coal. Most germanium is recovered during zinc or copper refining as a by-product. Germanium was extensively used in early semiconductor devices but has now been largely superseded by silicon. It is used as an alloying agent, catalyst, phosphor, and in infrared equipment. Symbol: Ge; m.p. 937.45°C; b.p. 2830°C; r.d. 5.323 (20°C); p.n. 32; r.a.m. 72.61.Опасность
Many of the chemicals used in the semiconductor industries are highly toxic. For example,germanium-halogen compounds are extremely toxic, both as a powder and in a gaseous state.Precautions should be taken when working with germanium as with similar metalloids fromgroup 14 (IVA).Промышленное использование
A rare elemental metal, germanium (Ge) has agrayish white crystalline appearance and hasgreat hardness: 6.25 Mohs. Its specific gravityis 5.35, and melting point is 937°C. It is resistantto acids and alkalies. It has metallic-appearingcrystals with diamond structure, givesgreater hardness and strength to aluminum andmagnesium alloys, and as little as 0.35% in tinwill double the hardness. It is not used commonlyin alloys, however, because of its rarityand great cost. It is used chiefly as metal inrectifiers and transistors. An Au–Ge alloy, withabout 12% germanium, has a melting point of359°C and has been used for soldering jewelry.Germanium is obtained as a by-productfrom flue dust of the zinc industry, or it can beobtained by reduction of its oxide from the ores,and is marketed in small irregular lumps. Germaniumcrystals are grown in rods up to 3.49cm in diameter for use in making transistorwafers. High-purity crystals are used for both P and Nsemiconductors. They are easier topurify and have a lower melting point than othersemiconductors, specifically silicon.
Возможный контакт
Because of its semiconductor proper ties, germanium is widely used in the electronic industry in rectifiers, diodes, and transistors. It is alloyed with alumi num, aluminum magnesium, antimony, bronze, and tin to increase strength, hardness, or corrosion resistance. In the process of alloying germanium and arsenic, arsine may be released; stibine is released from the alloying of germanium and antimony. Germanium is also used in the manufacture of optical glass for infrared applications; red-fluorescing phosphors; and cathodes for electronic valves; and in elec troplating; in the hydrogenation of coal; and as a catalyst, particularly at low temperatures. Certain compounds are used medically. Industrial exposures to the dust and fumes of the metal or oxide generally occur during separation and purification of germanium, welding, multiple-zone melting operations, or cutting and grinding of crystals. Germanium tetrahydride (germanium hydride, germane, and monoger mane) and other hydrides are produced by the action of a reducing acid on a germanium alloy.Экологическая судьба
Metals are recalcitrant to degradation; therefore, no biodegradation studies have been performed on germanium compounds. Naturally occurring germanium exists in mineral ores; therefore, the levels of free germanium are expected to be low and of low concern for bioaccumulation in aquatic and terrestrial species, due to negligible exposures.Перевозки
UN3089 Metal powders, flammable, n.o.s., Hazard Class: 4.1; Labels: 4.1-Flammable solid. UN1759 Corrosive solids, n.o.s., Hazard class: 8; Labels: 8-Corrosive material, Technical Name required.Методы очистки
Copper contamination on the surface and in the bulk of single crystals of Ge can be removed by immersion in molten alkali cyanide under N2. The Ge is placed in dry K and/or Na cyanide powder in a graphite holder in a quartz or porcelain boat. The boat is then inserted into a heated furnace which, after a suitable time, is left to cool to room temperature. At 750o, a 1mm thickness of metal requires about 1minute, whereas 0.5cm needs about half hour. The boat is removed from the furnace, and the solid samples are taken out with plastic-coated tweezers, carefully rinsed in hot water and dried in air [Wang J Phys Chem 60 45 1956, Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 712 1963]. Care with the use of cyanide.Несовместимости
A strong reducing agent and flammable solid. Finely divided metal is incompatible with oxidizing and nonoxidizing acids, ammonia, bromine, oxidizers, aqua regia, sulfuric acid, carbonates, halogens, and nitrates. Explosive reaction or ignition with potassium chlorate, potassium nitrate, chlorine, bromine, oxygen, and potas sium hydroxide in the presence of heat. Violent reaction with strong acids: aqua regia, nitric, and sulfuric. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions.Утилизация отходов
Recovery and return to sup pliers for reprocessing is preferable.Германий запасные части и сырье
сырьё
1of2
запасной предмет
Германий поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия |
|---|---|---|---|---|
| +86-17736087130 +86-18633844644 |
China | 994 | 58 | |
| +86-0371-55170693 +86-19937530512 |
China | 21632 | 55 | |
| +86-0371-86658258 +8613203830695 |
China | 29871 | 58 | |
| +86 319 5273535 | CHINA | 287 | 58 | |
| 86-13657291602 | CHINA | 22963 | 58 | |
| +86-023-6139-8061 +86-86-13650506873 |
China | 39894 | 58 | |
| +86-0551-65418671 +8618949823763 |
China | 34563 | 58 | |
| +86-029-89586680 +86-18192503167 |
China | 7724 | 58 | |
| 02120970332; +8613524231522 |
China | 2904 | 58 | |
| +8615250961469 | China | 9720 | 58 |